Naming Ionic and Molecular Compounds | How to Pass Chemistry
TLDRThe video script by Melissa Maribel offers a comprehensive guide on naming ionic and covalent (molecular) compounds, a crucial skill for chemistry students. It emphasizes the importance of identifying the type of compound first, whether ionic, consisting of a metal and nonmetal, or covalent, made up of two nonmetals. The script outlines strategies for naming ionic compounds, including those without transition metals, with transition metals, and those with polyatomic atoms. It also explains how to balance charges in ionic compounds and provides examples of naming and interpreting compound formulas. For covalent compounds, the focus is on prefixes and the composition of two nonmetals without charge balancing. The summary encourages students to practice and reassures them that with the right approach, they can excel in chemistry and achieve their career goals.
Takeaways
- 📚 Start by identifying the type of compound: ionic (metal and nonmetal) or molecular (covalent, two nonmetals).
- 🔍 For ionic compounds without transition metals, name the metal first, then the nonmetal ending in 'ide'.
- ⚖️ Balance charges in ionic compounds by using subscripts; memorize common charges for elements.
- 📐 Use Roman numerals in parentheses to indicate the charge of transition metals in ionic compounds.
- 🏷️ Polyatomic ions (like phosphate) are named without the nonmetal ending in 'ide' and have specific charges.
- 🌟 Memorize common polyatomic ions and their charges for proper compound naming.
- 🧩 When reversing the process from formula to name, use the charges to determine the correct Roman numeral for transition metals.
- 🔢 Covalent compounds use prefixes (1-10) and do not require charge balancing.
- 📝 The setup for covalent compounds is prefix + nonmetal name, followed by another prefix + 'ide' ending for the second nonmetal.
- ✍️ Practice is key to mastering compound naming; use flashcards and online resources for assistance.
- 📈 The secret to learning anything is persistence and the willingness to practice and understand the material.
- 📚 Online tutoring can provide additional help for those struggling with compound naming and chemistry concepts.
Q & A
What is the main focus of the video?
-The video focuses on teaching the different types of combinations for naming ionic and molecular (covalent) compounds.
What is the difference between ionic and molecular compounds?
-Ionic compounds consist of one metal and one nonmetal, while molecular (covalent) compounds consist of two nonmetals.
How do you identify the charges for aluminum and oxygen in aluminum oxide?
-Aluminum has a 3+ charge and oxygen has a 2- charge. These charges are balanced by placing a subscript of 2 next to aluminum and a subscript of 3 next to oxygen.
What does a Roman numeral in parentheses after a transition metal represent in an ionic compound?
-The Roman numeral represents the charge of the transition metal within the compound.
Why is it important to know polyatomic atoms when naming ionic compounds?
-Polyatomic atoms are groups of two or more elements that act as a single entity in a compound. Knowing them helps in correctly naming and balancing the charges in ionic compounds.
How do you balance charges in a compound with a polyatomic atom like calcium phosphate?
-You place the polyatomic group (phosphate) in parentheses and use subscripts to balance the total positive and negative charges, resulting in calcium phosphate.
What is the general structure for naming covalent compounds?
-The structure is a prefix indicating the number of atoms followed by the name of the nonmetal, and then another prefix followed by the name of the second nonmetal ending in 'ide'.
Why do covalent compounds not require balancing of charges?
-Covalent compounds consist of nonmetals that share electrons to form bonds, so there is no need to balance charges as there is no transfer of electrons as in ionic compounds.
What is the significance of prefixes in naming covalent compounds?
-Prefixes in covalent compounds indicate the number of atoms of each element present in the compound, and they do not involve balancing charges.
How do you determine the charge of a transition metal in a compound when given its formula?
-You can determine the charge by looking at the overall charges and using the fact that nonmetals like halogens typically have a -1 charge. The remaining charge goes to the transition metal.
What is the name of the compound with the formula FeBr2?
-The compound is named iron (II) bromide, where iron is the transition metal with a 2+ charge and bromine is the nonmetal with a -1 charge.
What is the name of the compound with the formula Cu3(PO4)2?
-The compound is named copper (II) phosphate, where copper is the transition metal with a 2+ charge and phosphate is the polyatomic ion with a 3- charge.
Outlines
📚 Introduction to Naming Compounds
This paragraph introduces the topic of the video, which is about learning how to name different types of ionic and covalent (molecular) compounds. The presenter, Melissa Maribel, emphasizes the importance of this lesson for students, as they will be naming compounds throughout their academic career in chemistry. The strategy for naming compounds is to first identify whether the compound is ionic or molecular. Ionic compounds are made of a metal and a nonmetal, while covalent compounds consist of two nonmetals. The periodic table is used to distinguish between metals and nonmetals. The paragraph also outlines the naming conventions for ionic compounds without transition metals, such as aluminum oxide, and provides the charges for aluminum and oxygen to demonstrate how to balance charges in ionic compounds.
🔬 Naming Ionic Compounds with Transition Metals and Polyatomic Ions
The second paragraph delves into the specifics of naming ionic compounds that contain transition metals and polyatomic ions. It explains the naming convention for ionic compounds with transition metals, which includes the metal's name, a Roman numeral in parentheses to indicate the metal's charge, and the nonmetal ending in 'ide'. An example given is iron(III) oxide, Fe2O3, where the Roman numeral 'III' indicates the charge of iron. The paragraph also covers polyatomic ions, which are groups of two or more elements acting as a single unit with a specific charge. It provides an example of calcium phosphate, illustrating how to balance the charges of calcium (Ca2+) and the phosphate ion (PO4^3-). The paragraph concludes with an example of an ionic compound with both a transition metal and a polyatomic ion, copper(II) nitrate, demonstrating how to balance the charges and use the correct subscripts.
🧪 Writing Names from Formulas and Covalent Compounds
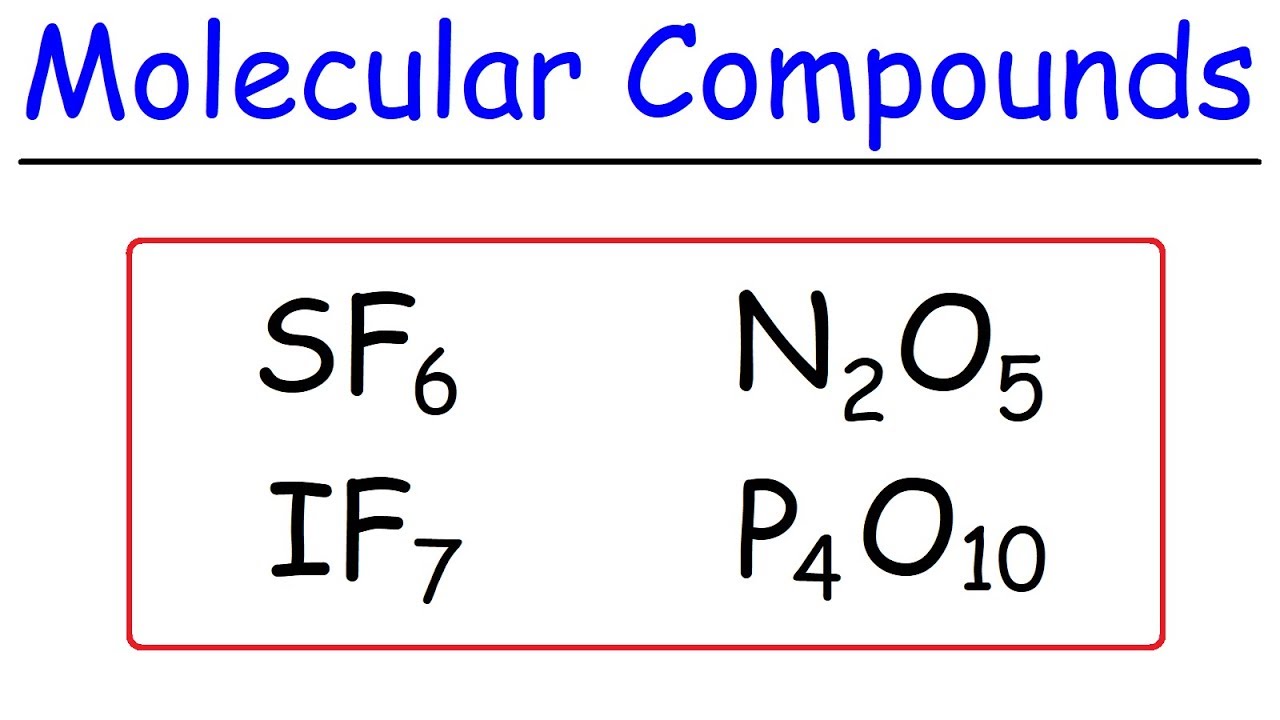
The third paragraph discusses how to write the names of compounds from their chemical formulas, specifically for ionic compounds. It uses FeBr2 and Cu3(PO4)2 as examples to show how to determine the charges and Roman numerals for transition metals, and how to identify the charges of polyatomic ions. The paragraph then transitions to covalent compounds, emphasizing the importance of knowing prefixes for the elements involved. It explains that covalent compounds do not require charge balancing and provides the naming convention, which involves prefixes followed by the nonmetal names ending in 'ide'. An example given is diphosphorus pentoxide, also known as phosphorus pentoxide, where 'di-' indicates two phosphorus atoms and 'pent-' indicates five oxygen atoms. The paragraph ends with an encouragement to practice the learned concepts and a reminder that understanding chemistry is achievable with effort and dedication.
Mindmap
Keywords
💡Ionic Compounds
💡Covalent Compounds
💡Transition Metals
💡Polyatomic Ions
💡Charge Balancing
💡Roman Numerals
💡Nonmetals
💡Metals
💡Prefixes
💡Chemical Formulas
💡Naming Conventions
Highlights
Introduction to the importance of learning how to name compounds in chemistry.
Differentiating between ionic and molecular (covalent) compounds as the first step in naming them.
Explanation of how to name ionic compounds without a transition metal, using Aluminum Oxide as an example.
Identifying and balancing charges for elements in ionic compounds, demonstrated with Aluminum and Oxygen.
Naming ionic compounds with a transition metal, including the use of Roman numerals to indicate charge.
The significance of knowing polyatomic atoms and their charges in naming ionic compounds.
Procedure for naming ionic compounds with polyatomic atoms, demonstrated with Calcium Phosphate.
Combining transition metals with polyatomic atoms in ionic compound names, using Copper (II) Nitrate as an example.
Method for determining the name of a compound from its formula, illustrated with FeBr2 and Cu3(PO4)2.
Understanding prefixes and their use in naming covalent compounds without balancing charges.
Constructing the name of covalent compounds using prefixes and the root names of nonmetals, exemplified by Diphosphorus Pentoxide.
The concept of 'Tri' and 'Hexa' prefixes in covalent compound nomenclature, demonstrated with N3O6.
Emphasis on practicing the learned concepts for better understanding and retention.
Offer of online tutoring for additional help, with a link provided in the description box.
Encouragement and motivation for learning and achieving career goals, emphasizing the secret to learning anything.
The periodic table's role in identifying metals and nonmetals for compound naming.
Use of parentheses and subscripts to balance charges in polyatomic ions within ionic compounds.
The naming convention for covalent compounds, focusing on prefixes and the absence of charge balancing.
Transcripts
Browse More Related Video

Naming Ionic Compounds with Transition Metals Introduction

How To Name Ionic Compounds With Transition Metals

Ionic vs. Molecular
How To Name Covalent Molecular Compounds - The Easy Way!
What Distinguishes Compounds from Molecules?
Elements, Atoms, Molecules, Ions, Ionic and Molecular Compounds, Cations vs Anions, Chemistry
5.0 / 5 (0 votes)
Thanks for rating: