Valence Bond Theory, Hybrid Orbitals, and Molecular Orbital Theory
TLDRProfessor Dave's video script delves into the concept of molecular orbitals, explaining how atomic orbitals hybridize to form molecular orbitals essential for covalent bonding. The script begins with a simple H2 molecule, illustrating how atomic orbitals overlap to create a sigma bond and form a molecular orbital shared by both electrons. It then discusses the formation of bonding and antibonding orbitals and the role of hybridization in multiple bond formation, using carbon as an example. The video also explains sp3, sp2, and sp hybridization and their implications for molecular orbitals. It further explores the creation of pi bonds from unhybridized p orbitals in double and triple bonds. The script concludes with a demonstration of how to generate and use orbital diagrams to calculate the bond order of molecules, using H2, O2, and N2 as examples. This comprehensive overview provides a clear understanding of molecular orbitals and their significance in chemical bonding.
Takeaways
- 🌟 **Atomic Orbitals Hybridization**: Atoms can hybridize their atomic orbitals into molecular orbitals, which are used to form covalent bonds through orbital overlap.
- 💥 **Sigma Bond Formation**: In H2, two hydrogen atoms form a sigma bond by overlapping their 1s atomic orbitals to create a molecular orbital for both electrons to reside in.
- 🔋 **Energy Conservation**: When two atomic orbitals form molecular orbitals, they create one bonding and one antibonding orbital, with electrons occupying the lower energy bonding orbital.
- 🔄 **Hybridization and Bonding**: An atom involved in multiple bonds uses hybrid orbitals; for example, carbon forms sp3 hybridized orbitals for four bonds, sp2 for three, and sp for two.
- 📈 **Energy Promotion**: To hybridize, carbon promotes an electron from the 2s to a 2p orbital, creating identical energy molecular orbitals for bonding.
- 🔬 **Bonding Behavior**: The number of bonds an atom can participate in determines the number of atomic orbitals that need to be hybridized to form the necessary molecular orbitals.
- 📊 **sp2 and sp Hybridization**: sp2 hybridization involves three electron domains (as in a double bond), while sp hybridization involves two electron domains (as in a triple bond).
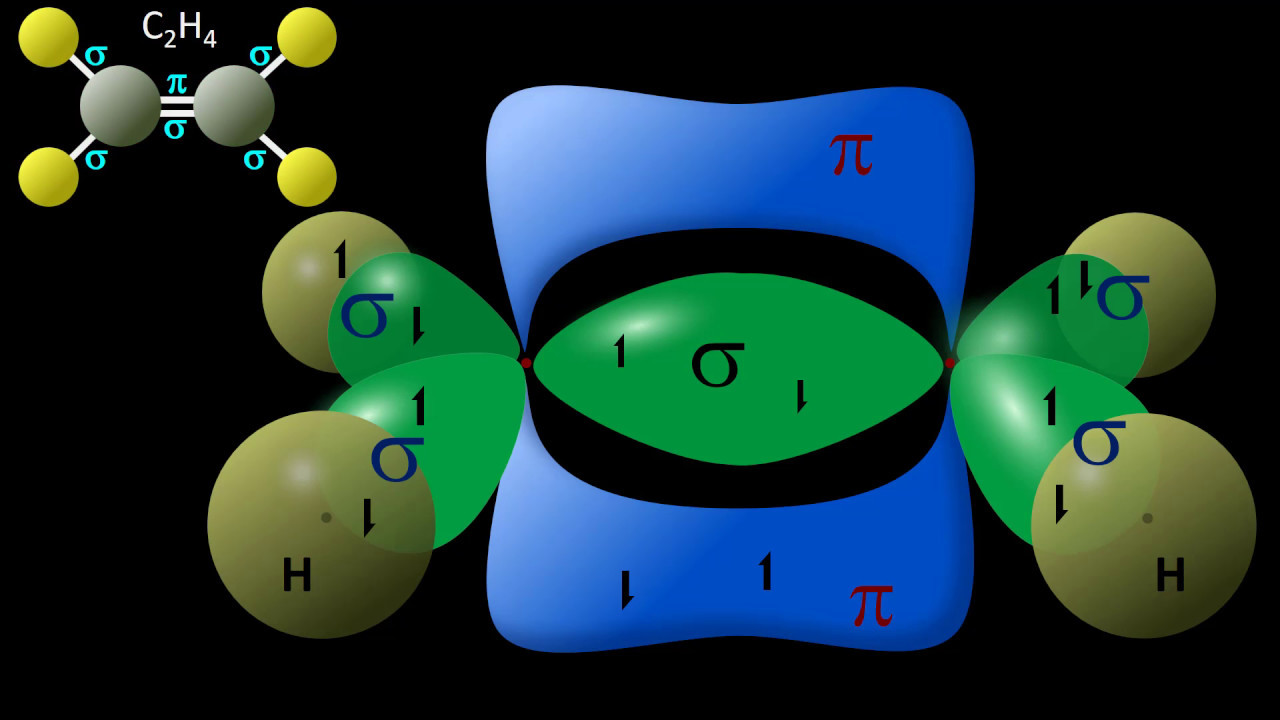
- 🛠️ **Pi Bond Formation**: Unhybridized p orbitals overlap to form pi bonds, which are found in double and triple bonds, extending perpendicularly from the molecular plane.
- 📐 **Orbital Diagrams**: Diagrams show atomic orbitals on the sides and molecular orbitals in between, used to calculate bond order and predict covalent bonding behavior.
- 🧮 **Bond Order Calculation**: The bond order is calculated as (number of electrons in bonding orbitals - number of electrons in antibonding orbitals) / 2.
- 🔬 **Molecular Stability**: The stability and type of bond (single, double, triple) in molecules like H2, O2, and N2 can be determined using orbital diagrams and bond order calculations.
Q & A
What is the significance of hybridization in molecular orbitals?
-Hybridization is significant as it allows an atom to combine its atomic orbitals to form hybrid orbitals that are used to make covalent bonds. This process is essential when an atom is participating in multiple bonds, as seen in carbon's sp3, sp2, or sp hybridization for forming four, three, or two bonds respectively.
How does the formation of a sigma bond in H2 occur?
-A sigma bond in H2 is formed when the 1s atomic orbitals of each hydrogen atom overlap, creating a molecular orbital that both electrons will reside in. This results in a lower energy state for the hydrogen molecule compared to the two separate hydrogen atoms due to the additional electrostatic interaction between the electrons and the opposite nuclei.
What happens when two atomic orbitals combine to form molecular orbitals?
-When two atomic orbitals combine, they form one bonding orbital and one antibonding orbital. The bonding orbital is at a lower energy level and is occupied by electrons, while the antibonding orbital is at a higher energy level and typically unoccupied unless the molecule is in an excited state.
How does the promotion of an electron in carbon allow for sp3 hybridization?
-To achieve sp3 hybridization, one of the 2s electrons in carbon is promoted to a vacant 2p orbital. This allows the 2s orbital and all three 2p orbitals to hybridize, creating four equivalent sp3 hybridized molecular orbitals that are degenerate in energy.
What is the role of unhybridized p orbitals in the formation of pi bonds?
-Unhybridized p orbitals, which are perpendicular to the plane of the molecule, overlap to form pi bonds. These are found in double and triple bonds, where the unhybridized p orbitals contain one electron each, and their overlap generates the pi bonds in addition to the sigma bond formed by hybridized orbitals.
How does the bond order in a molecule relate to the number of electrons in bonding and antibonding orbitals?
-The bond order is calculated by taking the difference between the number of electrons in bonding orbitals and the number of electrons in antibonding orbitals, then dividing by two. This value indicates the number of bonds between a pair of atoms in a molecule.
Why does an oxygen molecule (O2) have a double bond between its atoms?
-The bond order calculation for O2 is (10-6)/2, which equals 2. This is because the oxygen molecule has full sigma and pi orbitals, and half-filled pi* (antibonding) orbitals, resulting in a double bond between the oxygen atoms.
What is the difference between sp2 and sp hybridization in terms of electron domains and bond formation?
-An sp2 hybridized carbon has three electron domains, which can occur when it forms a double bond. It uses its 2s orbital and two of the 2p orbitals to form sp2 hybridized orbitals, leaving one unhybridized p orbital for pi bond formation. An sp hybridized carbon has two electron domains, typically seen in triple bond formations, and uses the 2s orbital and one 2p orbital to form sp hybridized orbitals, leaving two unhybridized p orbitals for two pi bonds.
What is the orientation of the unhybridized 2p orbitals in a molecule with a triple bond, such as C2H2?
-In a molecule with a triple bond like C2H2, the unhybridized 2p orbitals have a propeller-like orientation. They extend perpendicularly from the plane formed by the sp hybridized orbitals and overlap to form two pi bonds, in addition to the sigma bond formed by the overlap of the sp orbitals.
How can orbital diagrams be used to predict the covalent bonding behavior of any two atoms?
-Orbital diagrams can predict the covalent bonding behavior by showing the atomic orbitals of each atom and the resulting molecular orbitals after bond formation. By filling the molecular orbitals with electrons in the order of increasing energy, one can calculate the bond order and determine the type of bond (single, double, or triple) between the two atoms.
Why do nitrogen molecules have a triple bond between their atoms?
-The bond order calculation for nitrogen turns out to be 3, which is derived from the molecular orbital diagram and the distribution of electrons. This indicates that each nitrogen atom is involved in a triple bond with the other, consisting of one sigma bond and two pi bonds.
Outlines
🌟 Understanding Molecular Orbitals and Hybridization
Professor Dave introduces the concept of molecular orbitals, explaining how atomic orbitals can hybridize to form molecular orbitals which are used to create covalent bonds. He uses the example of H2 to illustrate the formation of a sigma bond through the overlap of atomic orbitals to form a molecular orbital. The lecture discusses the energy states of molecular orbitals, the role of electron configuration, and how the number of orbitals must be conserved, leading to the formation of both bonding and antibonding orbitals. The process of hybridization is further explained through the promotion of electrons and the combination of atomic orbitals to form hybrid orbitals, exemplified by sp3 hybridization in carbon making four bonds. The paragraph also touches on sp2 and sp hybridization, the formation of pi bonds, and the representation of these concepts through orbital diagrams. Finally, the importance of calculating bond order for determining the type of covalent bond present in molecules is covered.
🔬 Bond Order Calculation and Orbital Diagrams
This section delves into the calculation of bond order using molecular orbitals, which is defined as the difference between the number of electrons in bonding and antibonding orbitals divided by two. The example of the H2 molecule is used to demonstrate a single covalent bond, with a bond order of one. The explanation then moves on to O2, detailing the electron configuration and how the molecular orbitals are filled, leading to a bond order of two, indicative of a double bond. The concept is further applied to nitrogen, where the bond order calculation results in three, explaining the presence of a triple bond in a nitrogen molecule. The paragraph emphasizes the predictive power of orbital diagrams for determining covalent bonding behavior between atoms and encourages viewers to apply these concepts to understand molecular structures.
Mindmap
Keywords
💡Molecular Orbitals
💡Hybridization
💡Sigma Bond
💡Antibonding Orbital
💡Sp3 Hybridization
💡Sp2 Hybridization
💡Pi Bond
💡Bond Order
💡Atomic Orbitals
💡Covalent Bond
💡Orbital Diagrams
Highlights
Molecular orbitals are formed when atomic orbitals hybridize, creating shapes that resemble combinations of the original atomic orbitals.
Covalent bonds are made through the overlapping of these hybridized orbitals, as seen in the formation of a hydrogen molecule (H2).
The formation of molecular orbitals from atomic orbitals results in one bonding and one antibonding orbital, with electrons occupying the lower energy bonding orbital.
An electron can be excited from a bonding orbital to an antibonding orbital, which is key to understanding hybridization.
Carbon, when forming four bonds, uses sp3 hybridization, which involves promoting an electron and combining the 2s and three 2p orbitals into four identical hybrid orbitals.
The number of bonds an atom can form determines the hybridization needed to create the necessary molecular orbitals.
An sp2 hybridized carbon, involved in a double bond, uses three hybridized orbitals and one unhybridized p orbital for pi bond formation.
In a triple bond, as in C2H2, each carbon is sp hybridized, utilizing one 2s and one 2p orbital, leaving two unhybridized p orbitals for pi bond formation.
The linear molecule structure of C2H2 results from the propeller-like orientation of the unhybridized 2p orbitals forming two pi bonds beyond the sigma bond.
Orbital diagrams can be used to calculate the bond order of a molecule, which is the number of electrons in bonding orbitals minus those in antibonding orbitals, divided by two.
The bond order calculation for O2 results in a value of 2, indicating a double bond between the oxygen atoms.
Nitrogen molecules have a bond order of 3, which explains the triple bond between nitrogen atoms.
Orbital diagrams can predict the covalent bonding behavior of any two atoms, providing a visual and mathematical framework for understanding molecular structures.
The conservation of orbitals is a fundamental principle when atomic orbitals combine to form molecular orbitals.
The energy level of an atom dictates the types of orbitals available (s, p, d), influencing the hybridization process.
Hybridization allows for the formation of various bond types, including sigma and pi bonds, which are crucial for understanding molecular geometry and reactivity.
Excited electrons can move to higher energy orbitals, affecting the molecule's stability and reactivity.
The promotion of electrons is a necessary step in the hybridization process, particularly for atoms forming multiple bonds.
Transcripts
Browse More Related Video

1.3 Valence Bond Theory and Hybridization | Organic Chemistry

1.4 Molecular Orbital Theory | Organic Chemistry

Hybridization of Atomic Orbitals - Sigma & Pi Bonds - Sp Sp2 Sp3

13. Molecular Orbital Theory
Hybrid Orbitals explained - Valence Bond Theory | Orbital Hybridization sp3 sp2 sp

Lec-14 I Hybridization I Applied Chemistry I Chemical Engineering
5.0 / 5 (0 votes)
Thanks for rating: