General Chemistry Review for Organic Chemistry Part 2
TLDRIn this chemistry review, Melissa Maribel explores polarity in molecular structures, highlighting the importance of symmetry, electronegativity, and lone pairs in determining if a molecule is polar or nonpolar. She explains how electronegativity trends on the periodic table and emphasizes the significance of VSEPR theory for visualizing 2D structures in 3D, using examples like CH4 and CH3OH to demonstrate the process. The video sets the stage for further discussions on formal charges, resonance, and hybridization, crucial concepts for organic chemistry.
Takeaways
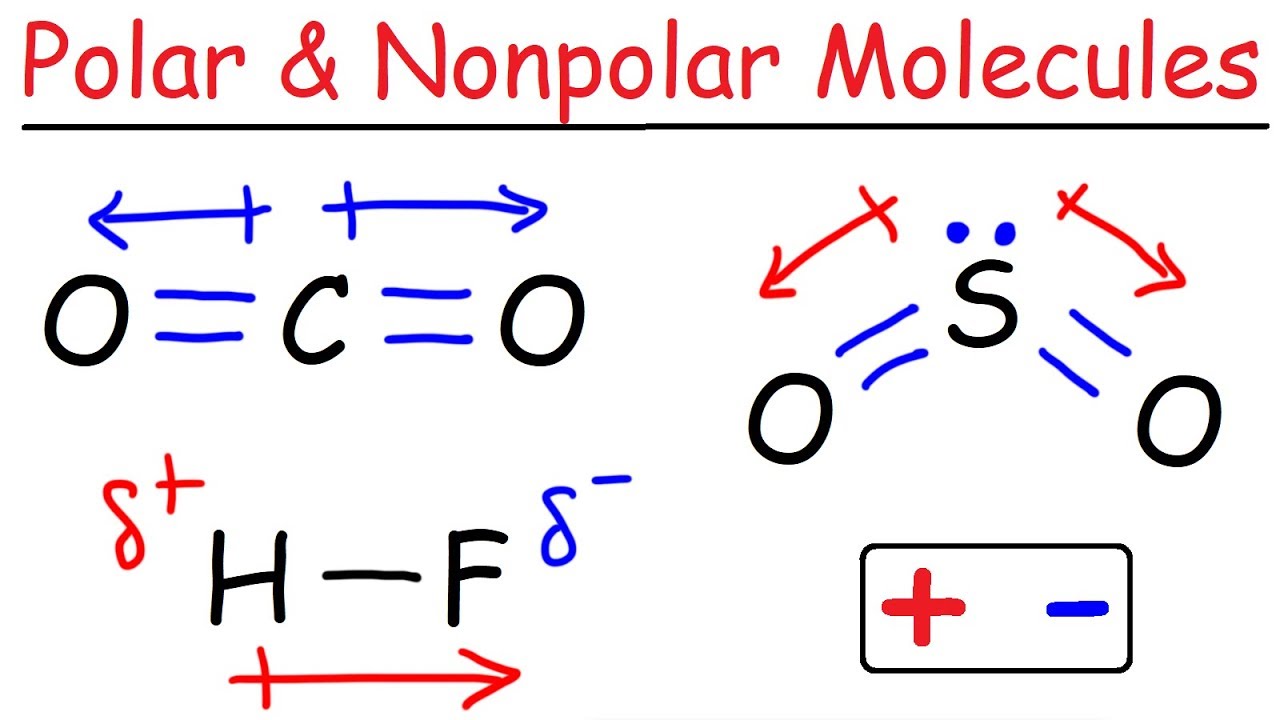
- 🧲 Polarity is determined by a tug-of-war between elements, where an unequal electron pull indicates a polar molecule.
- 📊 Symmetry is a key factor in polarity; symmetrical structures are nonpolar due to equal electron distribution, while asymmetrical structures are likely polar.
- 🌟 Electronegativity is crucial in polarity; it measures an atom's ability to attract electrons, with fluorine being the most electronegative.
- 📚 The periodic table provides a trend for electronegativity, which increases from left to right and up a group.
- 🌐 Noble gases are not electronegative as they are satisfied with their eight valence electrons and do not seek to attract more.
- 🔍 Comparing elements like oxygen and chlorine reveals that the higher element in the periodic table is more electronegative.
- 📐 Lone pairs on the central atom contribute to polarity, as seen in NH3 where the lone pair on nitrogen leads to an unequal electron distribution.
- 📈 VSEPR theory is essential for visualizing 2D structures in 3D, aiding in the understanding of molecular geometry.
- 📐 VSEPR form uses wedged, dashed, and solid lines to represent bonds coming towards you, hidden in the back, and on the plane, respectively.
- 🌐 The example of C2H6 demonstrates a tetrahedral geometry in its VSEPR form, illustrating the 3D structure of molecules.
- 🧪 CH3OH's Lewis structure and VSEPR form show the molecular geometry of carbon as tetrahedral and oxygen as bent, highlighting the importance of lone pairs in polarity.
Q & A
What is polarity in the context of chemistry?
-Polarity in chemistry refers to the unequal distribution of electron density between atoms in a molecule, which can be described as a tug-of-war between elements. If one side is stronger, it's polar, and if both sides are of equal strength, it's nonpolar.
What is a dipole moment and why is it significant in polar structures?
-A dipole moment is an unequal pull of electrons within a molecule, indicating the presence of polarity. It's significant because it helps to determine the directional nature of the electrostatic interactions in polar molecules.
What are the three main factors that determine the polarity of a molecule?
-The three main factors determining the polarity of a molecule are symmetry, electronegativity, and the presence of lone pairs on the central atom.
How does symmetry affect the polarity of a molecule?
-If a molecule has a symmetrical structure, it is likely to be nonpolar because there is an equal pull of electrons. However, if the structure is non-symmetrical, it is most likely polar due to an unequal pull of electrons.
What is electronegativity and how does it influence polarity?
-Electronegativity is a measure of an atom's ability to attract electrons. It influences polarity by determining which atom in a bond is more likely to 'win' the tug-of-war for electrons, thus creating a polar bond.
How does the position of an element on the periodic table relate to its electronegativity?
-Electronegativity generally increases as you move from left to right across a period and decreases as you move down a group on the periodic table. Elements closer to fluorine, which is the most electronegative, tend to be more electronegative themselves.
Why are noble gases not considered electronegative?
-Noble gases are not considered electronegative because they already have a full complement of eight valence electrons, making them stable and disinterested in attracting more electrons.
What is the significance of lone pairs on the central atom in determining polarity?
-Lone pairs on the central atom contribute to the polarity of a molecule. They can cause an unequal distribution of electron density, even in symmetrical molecules, making them polar.
What is VSEPR theory and how does it help in understanding molecular geometry?
-VSEPR (Valence Shell Electron Pair Repulsion) theory is a model used to predict the shapes of molecules based on the repulsion between electron pairs around a central atom. It helps in visualizing 2D structures in 3D, which is crucial for understanding molecular geometry.
How does the VSEPR theory apply to the molecule CH4?
-For CH4 (methane), the VSEPR theory predicts a tetrahedral geometry because the central carbon atom has four bonds with hydrogen atoms, and the electron pairs repel each other to occupy the corners of a tetrahedron.
Can you provide an example of how to visualize the 3D structure of C2H6 using VSEPR theory?
-C2H6 (ethane) has a tetrahedral geometry for each carbon atom. Using VSEPR theory, we can visualize the 3D structure by placing hydrogens around each carbon atom with one in the plane (solid line), one hidden in the back (dashed line), and one coming towards us (wedged line), and then bonding the two carbons together.
How does the molecular geometry of CH3OH differ from CH4 and C2H6?
-CH3OH (methanol) has a central carbon atom with a tetrahedral geometry, but the oxygen atom it is bonded to has a bent geometry due to the presence of two lone pairs, which affect the shape and polarity of the molecule.
Outlines
🔬 Polarity and Electronegativity in Chemistry
This paragraph introduces the concept of polarity in chemistry, comparing it to a tug-of-war between elements. It explains that polarity arises when there's an unequal pull of electrons, resulting in a dipole moment. The paragraph delves into three main factors affecting polarity: symmetry, electronegativity, and the presence of lone pairs on the central atom. It emphasizes the importance of electronegativity, which indicates an atom's ability to attract electrons, and how it varies across the periodic table. The paragraph uses examples to illustrate these concepts, including the comparison between oxygen and chlorine, and the impact of lone pairs on the polarity of molecules like NH3. It also introduces the VSEPR theory, which helps visualize 2D structures in 3D, using CH4 and C2H6 as examples to demonstrate how to draw molecular geometries in their 3D forms.
📚 Understanding Molecular Geometries with VSEPR Theory
This section continues the discussion on molecular polarity and introduces the concept of VSEPR (Valence Shell Electron Pair Repulsion) theory, which is crucial for visualizing 2D structures in 3D. The paragraph provides a step-by-step guide on how to draw the VSEPR form of molecules like CH4 and C2H6, explaining the use of wedged, dashed, and solid lines to represent different spatial orientations of atoms. It then moves on to CH3OH, combining the Lewis structure from a previous lesson with the molecular geometry to illustrate the bent shape due to the presence of lone pairs on the oxygen atom. The paragraph concludes by emphasizing the importance of understanding formal charges, resonance, and hybridization in organic chemistry, which are topics to be covered in the next part of the review.
Mindmap
Keywords
💡Polarity
💡Dipole Moment
💡Symmetry
💡Electronegativity
💡Fluorine
💡Noble Gases
💡Lone Pairs
💡VSEPR Theory
💡Tetrahedral Geometry
💡Bent Molecular Geometry
💡Formal Charges
💡Resonance
💡Hybridization
Highlights
Introduction to part two of the general chemistry review for organic chemistry by Melissa Maribel.
Review of Lewis structures from part one.
Explanation of polarity as a tug-of-war between elements in a molecule.
Polar molecules have a dipole moment due to an unequal pull of electrons.
Three main factors for determining polarity: symmetry, electronegativity, and lone pairs.
Symmetry in molecular structures can indicate nonpolar molecules.
Electronegativity defines an atom's ability to attract electrons and its trend across the periodic table.
Fluorine is identified as the most electronegative atom.
Noble gases are not electronegative due to their complete valence electron shells.
Comparison of electronegativity between oxygen and chlorine, with oxygen being more electronegative.
Determination of the most polar structure based on proximity to fluorine and electronegativity.
The impact of lone pairs on the central atom in increasing polarity.
Introduction to VSEPR theory for visualizing 2D structures in 3D.
CH4 VSEPR form demonstration with wedged, dashed, and solid lines.
C2H6 VSEPR form and 3D visualization with tetrahedral geometry.
CH3OH Lewis structure and VSEPR form with molecular geometry of bent for oxygen.
3D visualization of CH3OH structure incorporating both carbon and oxygen geometries.
Anticipation of part three covering formal charges, resonance, and hybridization in organic chemistry.
Transcripts
Browse More Related Video

Polarity, Resonance, and Electron Pushing: Crash Course Organic Chemistry #10

Resonance Structures/Assigning Formal Charge
Polar and Nonpolar Molecules

Intermolecular Forces and Trends, Formal Charges, Hund's Rule, Lattice Structures and Unit Cells

8.5 Molecular Polarity | High School Chemistry

1.1 Lewis Structures | Organic Chemistry Complete Course
5.0 / 5 (0 votes)
Thanks for rating: