Atomic Structure full topic
TLDRThe video script delves into atomic structure, focusing on the concept of atoms, their composition, and the significance of the periodic table of elements. It explains the role of subatomic particles like neutrons, protons, and electrons, and how they contribute to an atom's properties. The script further explores the structure of an atom, detailing the nucleus and electron shells, and introduces the concept of quantum numbers and orbitals. The discussion also touches on the photoelectric effect, Planck's quantum theory, and the dual wave-particle nature of light. The script emphasizes the importance of understanding atomic structure and electron configurations, providing foundational knowledge for further studies in chemistry and physics.
Takeaways
- 🔬 Atoms are the smallest particles of an element that retain the element's properties.
- 💫 An atom consists of protons, neutrons, and electrons, with protons being positively charged, electrons negatively charged, and neutrons neutral.
- ⚫️ In a neutral atom, the number of protons equals the number of electrons.
- 📊 The periodic table organizes elements like sodium and oxygen, with each element having unique properties based on its atomic structure.
- 🌐 Electrons are arranged in shells or energy levels around the nucleus, with each shell having a specific capacity for electrons based on the formula 2n^2.
- 🔄 Quantum numbers (n, l, m, s) describe an electron's energy level, orbital shape, orientation, and spin direction within an atom.
- 🌌 The Pauli Exclusion Principle states that no two electrons in an atom can have the same set of quantum numbers due to differences in their magnetic spin quantum number.
- 🌈 The photoelectric effect involves the emission of electrons from a metal surface when struck by light of a certain frequency, with no emission below the threshold frequency.
- 💥 The energy of a photon is directly proportional to its frequency, as described by Planck's quantum theory and the equation E = hν, where h is Planck's constant and ν is the frequency.
- 📈 The wave-particle duality of light explains how light exhibits both wave-like and particle-like properties, such as interference and photoelectric effects.
- 🚧 The limitations of the Bohr atomic model include its inability to explain certain phenomena like the Zeeman effect and the Stark effect, and its violation of the Heisenberg uncertainty principle.
Q & A
What is an atom and how is it defined?
-An atom is defined as the smallest particle of an element that retains all the properties of that element. It is made up of subatomic particles like neutrons, protons, and electrons.
What are the three subatomic particles that make up an atom and what are their charge properties?
-The three subatomic particles that make up an atom are neutrons, protons, and electrons. Neutrons have no charge and are neutral, protons have a positive charge, and electrons have a negative charge.
How does the structure of an atom relate to its electron configuration?
-The structure of an atom is closely related to its electron configuration. Electrons are arranged in shells or energy levels around the nucleus, and each shell can hold a specific number of electrons based on the formula 2n^2, where n is the energy level.
What is the significance of the periodic table of elements in relation to atoms?
-The periodic table of elements organizes elements based on their atomic number, which is the number of protons in an atom's nucleus. This arrangement helps to predict the properties and behavior of elements and their atoms.
How does the Bohr atomic model contribute to our understanding of atomic structure?
-The Bohr atomic model introduces the concept of electrons orbiting the nucleus in defined circular paths or shells. It helps explain the quantization of energy levels and the emission or absorption of energy in discrete quantities when electrons transition between these levels.
What are the limitations of the Bohr atomic model?
-The Bohr atomic model fails to explain certain phenomena such as the Zeeman effect and the Stark effect. It also violates the Heisenberg uncertainty principle, which states that it is impossible to simultaneously know the exact position and momentum of a particle.
What is the photoelectric effect and how does it relate to the properties of light?
-The photoelectric effect is the emission of electrons from a metal surface when it is struck by light of a certain frequency. It demonstrates that light has particle-like properties, as the emission of electrons occurs in discrete packets or quanta of energy, supporting the concept of light as both a wave and a particle.
What is Planck's quantum theory and how does it relate to the emission and absorption of energy by atoms?
-Planck's quantum theory states that energy is not emitted or absorbed continuously but in discrete quantities or quanta. This theory introduced the concept of the smallest amount of energy that can be emitted or absorbed, known as a quantum, and established a direct proportionality between the energy of radiation and its frequency.
How is the energy of a photon calculated using Planck's constant and the frequency of light?
-The energy of a photon is calculated using the formula E = hν, where E is the energy of the photon, h is Planck's constant, and ν (nu) is the frequency of the light. This equation shows that the energy of a photon is directly proportional to the frequency of the radiation.
What is the relationship between wavelength, frequency, and the speed of light?
-The relationship between wavelength (λ), frequency (f), and the speed of light (c) is given by the equation c = λν. This equation shows that the speed of light is equal to the product of the wavelength and the frequency of the wave.
What is the significance of the wave-particle duality in quantum mechanics?
-The wave-particle duality is a fundamental concept in quantum mechanics that states that particles such as electrons and photons exhibit both wave-like and particle-like properties. This duality is crucial for understanding various quantum phenomena, including the photoelectric effect and the behavior of atoms.
Outlines
🧪 Introduction to Atoms and Atomic Structure
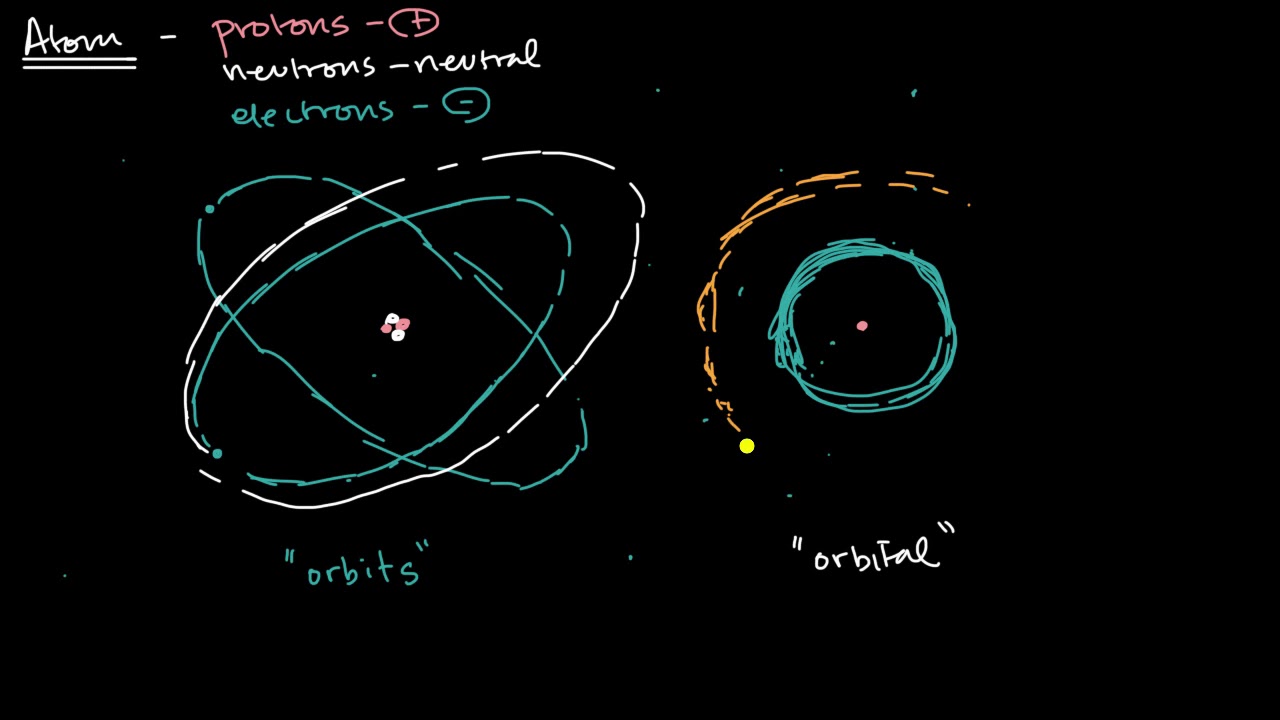
This paragraph introduces the concept of an atom, defining it as the smallest particle of an element with all the properties of that element. It explains the structure of an atom, comprising neutrons, protons, and electrons. The paragraph also discusses the importance of the periodic table of elements and the atomic number, which represents the number of protons in an atom. The key takeaway is understanding the neutral atom, where the number of electrons equals the number of protons, and the mass number, which represents the total number of protons and neutrons.
📊 Electron Configuration and Quantum Numbers
This paragraph delves into the electron configuration within an atom, explaining the significance of electron shells and subshells. It introduces the concept of quantum numbers, which are used to describe the state of an electron in an atom. The paragraph discusses the relationship between the number of electrons in each shell and the energy levels of those electrons. The explanation includes the formula for calculating the maximum number of electrons in each shell, which is dependent on the shell's energy level. The main points include understanding the distribution of electrons in an atom and the role of quantum numbers in defining electron states.
🌐 Atomic Models: Bohr and Quantum
This paragraph discusses the Bohr atomic model, highlighting its key postulates and limitations. It explains how electrons revolve around the nucleus in fixed orbits and that each orbit has a specific energy level. The paragraph also touches on the quantum mechanical model, emphasizing the probabilistic nature of electron locations. The limitations of the Bohr model, such as its inability to explain certain atomic phenomena, are also mentioned. The focus is on understanding the evolution of atomic models and the transition from Bohr's model to more advanced quantum theories.
🌊 Wave-Particle Duality and Planck's Quantum Theory
This paragraph explores the wave-particle duality of light, discussing how light exhibits both wave-like and particle-like properties. It introduces Planck's quantum theory, which posits that energy is emitted or absorbed in discrete quantities called quanta. The paragraph explains the relationship between the energy of a photon and its frequency, as well as the concept of the photoelectric effect. The key points include understanding the dual nature of light, the significance of quanta, and the implications of Planck's theory for atomic structure and energy transitions.
🔬 Atomic Spectra and Quantum Mechanics
This paragraph discusses the concept of atomic spectra, particularly the hydrogen spectrum, and how it relates to electron transitions within an atom. It explains the emission and absorption of light by atoms, and how these processes are described by quantum mechanics. The paragraph introduces the Rydberg formula for calculating the wavelengths of spectral lines and the significance of series limits. The main focus is on understanding how atomic spectra provide insights into the energy levels of atoms and the role of quantum mechanics in explaining these phenomena.
📚 Electron Transitions and the Role of Orbitals
This paragraph delves into the concept of electron transitions and the role of orbitals in atoms. It explains how electrons move between different energy levels, leading to the emission or absorption of photons. The paragraph introduces the idea of orbitals as regions of space where there is a high probability of finding an electron. It also discusses the different types of orbitals (s, p, d, f) and their capacities for electrons. The key points include understanding the process of electron transitions, the significance of orbitals in electron configuration, and how these concepts are applied in the study of atomic structure.
Mindmap
Keywords
💡Atom
💡Periodic Table
💡Protons
💡Electrons
💡Neutrons
💡Electron Configuration
💡Quantum Numbers
💡Energy Levels
💡Orbitals
💡Pauli Exclusion Principle
💡Atomic Structure
Highlights
An atom is defined as the smallest particle of an element that retains the properties of that element.
The periodic table of elements organizes elements like sodium and oxygen based on their properties.
An atom consists of neutrons, protons, and electrons.
In a neutral atom, the number of electrons is equal to the number of protons.
The atomic number represents the number of protons in an atom.
The mass number is the sum of the number of protons and neutrons in an atom's nucleus.
Electrons are arranged in shells or energy levels around the nucleus.
The closer an electron shell is to the nucleus, the lower its energy level.
The maximum number of electrons that each shell can hold follows the 2n^2 rule, where n is the shell number.
The electron configuration of an element can be represented using the orbitals (s, p, d, f).
The Bohr atomic model explains the distribution of electrons in discrete energy levels around the nucleus.
The photoelectric effect involves the emission of electrons from a metal surface when struck by light.
There is a threshold frequency below which no electrons are emitted, regardless of the light's intensity.
Above the threshold frequency, the intensity of light affects the number of electrons emitted.
The kinetic energy of emitted electrons increases with the frequency of the light above the threshold frequency.
Light exhibits both wave-like and particle-like properties, known as wave-particle duality.
The Planck's quantum theory states that energy is emitted or absorbed in discrete quantities, not continuously.
Transcripts
Browse More Related Video

Structure of the Atom - Proton, Neutron, Electron - Atomic Number & Mass Number - [1-2-6]

Cram AP Chem Unit 1: Atomic Structures and Properties

Atoms and Molecules (ionic vs covalent bonds)

Atoms | What are They? What are Protons, Neutrons and Electrons?
Shells, subshells, and orbitals | Atomic structure and properties | AP Chemistry | Khan Academy

Applied Chemistry_Atomic Charecteristics_ Lecture 06 for Polytechnic 1st Semester
5.0 / 5 (0 votes)
Thanks for rating: