Is Matter Around Us Pure Class 9
TLDRThis chemistry class focused on the concepts of elements, compounds, and mixtures, aiming to simplify these topics for students. The instructor began by defining matter and distinguishing it from non-matter, such as thoughts and emotions. They then explained that matter can be categorized into pure substances and mixtures based on chemical properties. Pure substances, which cannot be broken down into simpler substances by physical means, are further divided into elements (single type of atoms) and compounds (two or more elements chemically combined). Mixtures, containing two or more different kinds of particles, are either homogeneous (uniform throughout) or heterogeneous (non-uniform). Examples provided include gold as an element and water as a compound, with potassium permanganate solution and soil illustrating mixtures. The class encouraged interactive learning and practical understanding of these fundamental chemistry concepts.
Takeaways
- 🧪 Matter is defined in science as anything that has mass and volume and occupies space, such as water, chairs, tables, smoke, food, and juice.
- 🌌 Non-matter includes abstract concepts like thoughts and emotions, which do not have mass or volume and do not occupy space.
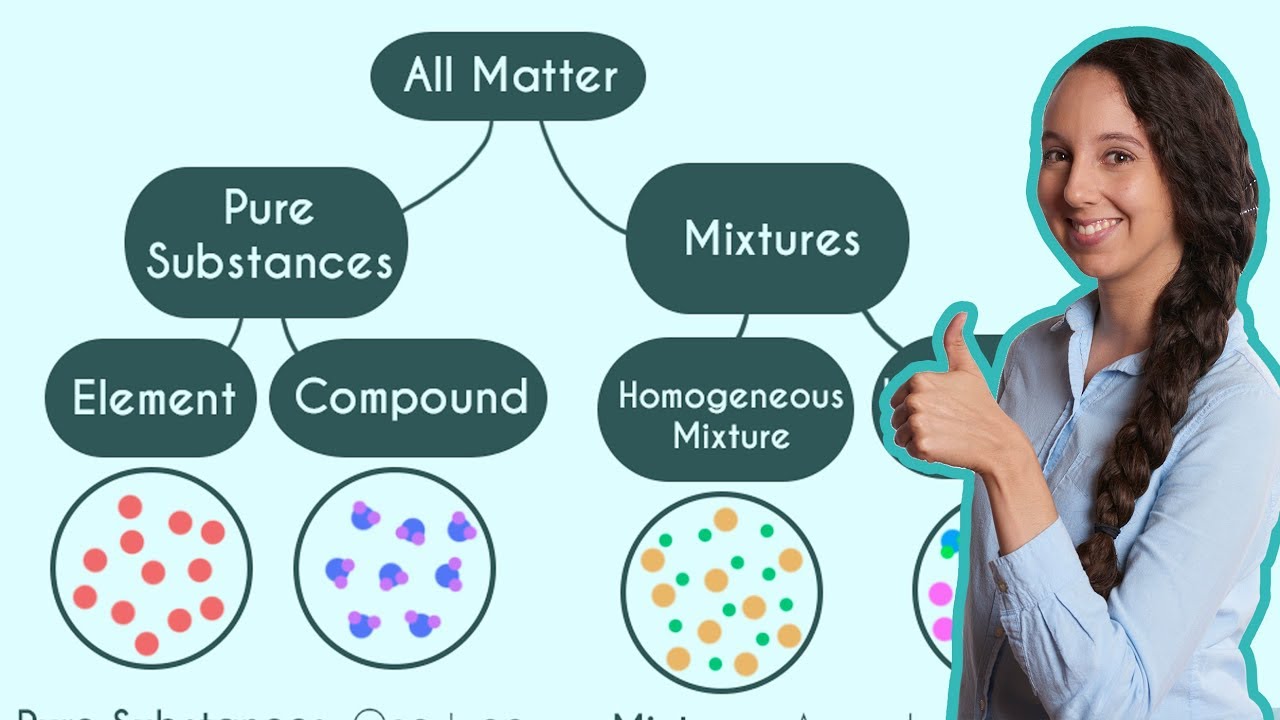
- 🔍 Matter can be classified based on its physical properties into solid, liquid, and gas, and chemically into pure substances and mixtures.
- 📚 A pure substance is made up of one kind of particle and cannot be separated into simpler substances by physical means, such as gold or water.
- 🧬 Pure substances can further be divided into elements, which contain only one type of atom, and compounds, which are made up of molecules containing more than one type of atom chemically combined, like water (H2O) and sugar (C12H22O11).
- 🔬 Elements are the simplest substances in chemistry that cannot be split into simpler substances by chemical means and contain only one kind of atom, such as gold (Au), iron (Fe), oxygen (O2), and hydrogen (H2).
- 🤝 Compounds are substances composed of two or more elements chemically combined in a fixed proportion, like sodium chloride (NaCl) and water (H2O).
- 🫕 Mixtures consist of two or more pure substances mixed together and can be separated into simpler substances by physical means, such as potassium permanganate solution and soil.
- 💧 Homogeneous mixtures, also known as solutions, have a uniform composition throughout, like sugar solution and aerated drinks, whereas heterogeneous mixtures have a non-uniform composition, like soil and oil and water.
- 🌳 The classification of milk and wood as either elements, compounds, or mixtures is a topic for further exploration, with milk being a tricky case that appears homogeneous but is actually heterogeneous.
Q & A
What are the three main states of matter?
-The three main states of matter are solid, liquid, and gas. These states are classified based on the physical properties of matter.
What is the difference between a pure substance and a mixture?
-A pure substance is made up of only one kind of particle and cannot be separated into simpler substances by physical means. A mixture, on the other hand, contains two or more different kinds of particles and can be separated by physical means.
How is matter classified based on chemical properties?
-Based on chemical properties, matter can be divided into pure substances and mixtures. Pure substances can further be classified into elements and compounds.
What is an element in chemistry?
-An element is a pure substance that cannot be split into simpler substances by chemical means and contains only one kind of atom.
What is a compound in chemistry?
-A compound is a substance composed of two or more elements that are chemically combined in a fixed proportion.
How many elements are there in the periodic table?
-There are 118 elements in the periodic table, as mentioned in the script.
What is a homogeneous mixture and what is an example?
-A homogeneous mixture is a uniform mixture that looks the same throughout. An example given in the script is a potassium permanganate solution, which appears uniformly purple throughout.
What is a heterogeneous mixture and what is an example?
-A heterogeneous mixture is a non-uniform mixture that has parts with different compositions. An example from the script is soil, which contains sand, silt, clay, and other particles in varying proportions.
Why are mixtures considered impure substances?
-Mixtures are considered impure substances because they contain two or more pure substances mixed together, resulting in a composition that is not uniform throughout.
What is the difference between a homogeneous and a heterogeneous mixture in terms of appearance?
-A homogeneous mixture has a uniform appearance throughout, whereas a heterogeneous mixture has a non-uniform appearance with different parts having different compositions.
Can mixtures have a fixed formula like compounds?
-No, mixtures do not have fixed formulas because they are not chemically combined in a fixed proportion. The composition of a mixture can vary.
What is the classification of milk in terms of mixtures?
-Milk is classified as a heterogeneous mixture. Although it appears homogeneous, it is actually a mixture of water, fats, and proteins that are not uniformly mixed.
What is the importance of understanding the difference between pure substances and mixtures?
-Understanding the difference between pure substances and mixtures is crucial in chemistry as it helps in the analysis and identification of the composition of various materials. It also aids in processes like purification and synthesis.
Outlines
🌟 Introduction to Elements, Compounds, and Mixtures
The video begins with a warm welcome and an introduction to the topic of matter, focusing on elements, compounds, and mixtures. The instructor emphasizes the importance of understanding these concepts in chemistry and encourages viewers to watch the entire class. The video also promotes the website Chaotica.me for courses in physics, chemistry, and upcoming math courses, highlighting current discounts. The audience is engaged by asking them to define matter and differentiate between matter and non-matter, using examples like a chair, water, and a shadow. The classification of matter based on physical properties into solid, liquid, and gas is briefly mentioned before diving into the chemical properties, distinguishing between pure substances and mixtures.
🔍 Understanding Pure Substances and Mixtures
This paragraph delves into the definition of a pure substance, which is made up of one kind of particle, and provides examples such as pure gold and water. The difference between physical and chemical separation methods is clarified, noting that pure substances cannot be separated into simpler substances by physical means. The concept of mixtures, containing two or more pure substances, is introduced with examples like potassium permanganate solution and soil. The instructor explains that mixtures can be separated into their constituent parts by physical means, contrasting them with pure substances, which are indivisible by such methods.
🔬 Elements and Compounds: The Building Blocks of Matter
The video continues by exploring the differences between elements and compounds. An element is defined as a pure substance consisting of only one type of atom, which cannot be broken down into simpler substances by chemical means. Examples given include gold (Au) and iron (Fe), which are both elements. Compounds, on the other hand, are substances composed of two or more elements that are chemically combined in a fixed proportion. Water (H2O) and sugar (C12H22O11) are used as examples of compounds, with the instructor emphasizing the fixed ratio of atoms in a compound. The distinction between elements, which are the simplest substances, and compounds, which are chemical combinations, is highlighted.
🧪 Classifying Pure Substances: Elements vs. Compounds
The audience is encouraged to classify substances as either elements or compounds based on their composition. Oxygen (O2) and hydrogen (H2) are confirmed as elements due to their consistence of a single type of atom. Salt (NaCl) and water (H2O) are identified as compounds because they consist of two different types of atoms chemically combined. Silver, with the symbol Ag, is also discussed as an element. The importance of understanding chemical formulas to determine if a substance is an element or a compound is stressed, as a compound will always consist of more than one type of atom.
🤔 Diving into Mixtures: Homogeneous vs. Heterogeneous
The video then focuses on mixtures, explaining that they contain two or more different kinds of particles and can be divided into homogeneous and heterogeneous mixtures. Homogeneous mixtures have a uniform composition throughout, exemplified by a potassium permanganate solution, which appears the same throughout. Heterogeneous mixtures, such as soil, have a non-uniform composition and can be differentiated into their components based on their physical properties. The distinction between the two types of mixtures is made clear through examples and the concept of uniformity.
🏗️ Practical Examples of Mixtures: Identifying Homogeneous and Heterogeneous
The instructor engages the audience with practical examples to determine whether common mixtures are homogeneous or heterogeneous. Oil and water are identified as a heterogeneous mixture due to their separation into layers when combined. A sugar solution in water is classified as homogeneous because the sugar dissolves uniformly. Aerated drinks, like soda, are also homogeneous due to the even distribution of carbon dioxide throughout. Lastly, iron and sulfur powder, when mixed, form a heterogeneous mixture because they do not mix uniformly and can be seen as separate components.
🌈 Summary of Matter Classification
The video concludes with a summary of the key concepts discussed. Matter is divided into pure substances and mixtures based on chemical properties. Pure substances, which contain only one type of particle, are further divided into elements and compounds. Elements are the simplest substances with a single type of atom, while compounds consist of chemically combined elements in a fixed proportion. Mixtures, containing more than one kind of particle, are classified as homogeneous (uniform throughout) or heterogeneous (non-uniform). The instructor also mentions that there are 118 known elements and encourages the audience to apply their knowledge to classify additional substances like milk and wood.
📚 Final Thoughts and Further Learning
The instructor encourages students to think critically and apply the concepts learned to classify wood as an element, compound, or type of mixture. They are prompted to share their answers in the comments section. The video ends with a reminder to visit the Manoj Academy website for further courses and discounts, and to subscribe to the YouTube channel for more informative content. The importance of continued learning and staying safe is emphasized, and the instructor expresses gratitude for the interactive participation in the session.
Mindmap
Keywords
💡Matter
💡Elements
💡Compounds
💡Mixtures
💡Homogeneous Mixtures
💡Heterogeneous Mixtures
💡Physical Properties
💡Chemical Properties
💡Pure Substances
💡Colloids
💡Wood
Highlights
Introduction to the chemistry class focusing on elements, compounds, and mixtures.
The definition of matter as something with mass and volume that occupies space.
Differentiating between matter and non-matter, such as thoughts and emotions.
Classification of matter into solids, liquids, and gases based on physical properties.
The distinction between pure substances and mixtures based on chemical properties.
Definition of a pure substance as one made up of a single kind of particle.
Examples of pure substances, including gold and water, explained.
Explanation of why pure substances cannot be separated by physical means.
Mixtures defined as containing two or more pure substances mixed together.
Examples of mixtures, such as potassium permanganate solution and soil.
The difference between elements and compounds within pure substances.
Elements are substances that cannot be split into simpler substances by chemical means.
Compounds are substances composed of two or more elements chemically combined.
The fixed proportion of elements in compounds, using water (H2O) and sugar (C12H22O11) as examples.
Differentiating between homogeneous and heterogeneous mixtures.
Homogeneous mixtures, also known as solutions, are uniform throughout.
Heterogeneous mixtures are non-uniform and can be separated by physical means.
Practical examples to distinguish between homogeneous and heterogeneous mixtures.
The importance of understanding the difference between pure substances and mixtures in chemistry.
Interactive session with students to classify various substances into elements, compounds, mixtures, and types of mixtures.
Summary of the key concepts learned in the class about matter classification.
Invitation for students to visit the academy's website for courses and discounts.
Transcripts
Browse More Related Video

Types of Matter - Elements, Compounds, Mixtures, and Pure Substances
Pure Substances and Mixtures! (Classification of Matter)

Pure Substances and Mixtures | Chemistry

What are Homogeneous & Heterogeneous Mixtures in Chemistry?

How to Compare Pure Substances and Mixtures - HSC Chemistry

Mixtures - Class 9 Tutorial
5.0 / 5 (0 votes)
Thanks for rating: