Ionization Energy Electron Affinity Atomic Radius Ionic Radii Electronegativity Metallic Character
TLDRThis video script offers an in-depth exploration of periodic trends, focusing on atomic and ionic radii, electronegativity, ionization energy, electron affinity, and metallic character. It explains how atomic size increases down a group and to the left across the periodic table, influenced by nuclear charge and electron shielding effects. The script delves into ionic radii, highlighting that cations are smaller than their neutral atoms due to lost electron shells, while anions are larger because of increased electron repulsion. Electronegativity is portrayed as increasing towards the upper right of the periodic table, with nonmetals being more electronegative than metals. Metallic character is shown to increase down a group and to the left across the periodic table, with metals more willing to lose electrons. Ionization energy, the energy needed to remove an electron, is detailed to increase across a period due to a stronger nuclear grip on electrons, though distance between the nucleus and valence electrons also plays a significant role. Electron affinity, the energy change when an electron is added to an atom, typically results in exothermic reactions for electronegative elements. The video script concludes with examples that illustrate these concepts, providing a comprehensive understanding of periodic trends.
Takeaways
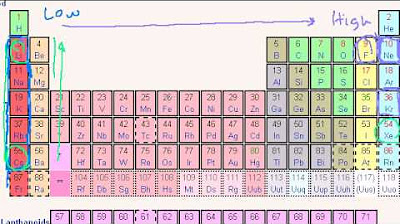
- 📊 **Atomic Radius Trends**: Atomic size increases as you move down a group and to the left across a period in the periodic table.
- 🚀 **Nuclear Charge vs. Distance**: The size of an atom is influenced more by the distance between the nucleus and electrons than by the atomic mass.
- 🪜 **Shells and Atomic Size**: Atoms with more electron shells are larger because the outer electrons are further from the nucleus.
- 🛡️ **Shielding Effect**: Inner core electrons shield outer electrons from the nucleus, reducing the effective nuclear charge experienced by valence electrons.
- ⚛️ **Ionic Radii Differences**: Cations are smaller than their parent atoms due to loss of an electron shell, while anions are larger because of increased electron repulsion.
- ➡️ **Electronegativity**: It increases across a period from left to right and decreases down a group, with nonmetals being more electronegative than metals.
- 🧲 **Metallic Character**: Metals, found on the left side of the periodic table, have a higher metallic character, meaning they tend to lose electrons more easily.
- ⚡ **Ionization Energy**: It is the energy required to remove an electron and generally increases across a period and decreases down a group due to increasing distance from the nucleus.
- 🔄 **Electron Affinity**: The energy change when an electron is added to an atom, which is usually exothermic for nonmetals and endothermic for noble gases and some alkaline earth metals.
- 📉 **Exceptions in Trends**: There are instances where ionization energy decreases despite an increase in nuclear charge due to electron shielding and orbital energy levels.
- 🔗 **Electron Configuration Stability**: Elements with half-filled or empty orbitals are more likely to release energy upon gaining an electron, making electron affinity more exothermic.
Q & A
Why is hydrogen considered larger than helium despite having a lower atomic mass?
-Hydrogen is considered larger than helium because it has a lower nuclear charge, which results in a weaker force of attraction between the nucleus and its electron, allowing the electron cloud to be more spread out. Helium, with a higher atomic mass and more protons, has a stronger nuclear charge attracting its electrons more tightly, causing the electron cloud to shrink and making its atomic radius smaller.
How does the atomic radius trend when moving down a group in the periodic table?
-The atomic radius increases as you move down a group in the periodic table. This is because each successive element adds an additional electron shell, which increases the distance between the nucleus and the outermost electrons, thus increasing the atomic size.
What is the effect of electron shielding on the atomic size?
-Electron shielding is the phenomenon where inner core electrons partially shield the outer valence electrons from the full effect of the positive charge of the nucleus. This shielding reduces the effective nuclear charge experienced by the outer electrons, which can result in a larger atomic size as the outer electrons are less tightly held by the nucleus.
How does the charge of an ion affect its size compared to its parent atom?
-A cation (positively charged ion) is significantly smaller than its parent atom because it has lost an electron and thus has less electron-electron repulsion. Conversely, an anion (negatively charged ion) is larger than its parent atom due to the addition of electrons, which increases electron-electron repulsion and causes the electron cloud to expand.
Why is the ionization energy generally higher for nonmetals than for metals?
-Nonmetals have a higher ionization energy because they tend to gain electrons to achieve a stable electron configuration, making it more difficult to remove an electron from them. Metals, on the other hand, have a lower ionization energy as they tend to lose electrons to achieve stability, so it is easier to remove an electron from a metal.
How does the ionization energy trend within a period (row) of the periodic table?
-The ionization energy generally increases from left to right across a period. This is due to an increase in the effective nuclear charge as you move across the row, which results in a stronger attraction between the nucleus and the valence electrons, making them harder to remove.
What is the relationship between electronegativity and the position of an element on the periodic table?
-Electronegativity increases from left to right and decreases from top to bottom on the periodic table. Nonmetals, found on the right side of the periodic table, are more electronegative as they have a greater tendency to attract electrons towards them, while metals, on the left side, are less electronegative and more likely to lose electrons.
How does the metallic character of elements change as you move down a group in the periodic table?
-The metallic character increases as you move down a group in the periodic table. This is because the elements become larger and the outer electrons are further from the nucleus, making them easier to lose, which is a characteristic behavior of metals.
What is the general trend for electron affinity as you move across a period on the periodic table?
-Electron affinity generally increases from left to right across a period. This is because elements on the right side of the periodic table are more electronegative and have a stronger attraction for additional electrons, leading to a more exothermic electron affinity.
Why is the addition of an electron to noble gases (group 8) typically endothermic?
-Adding an electron to noble gases is typically endothermic because their outer electron shells are completely filled, resulting in a stable configuration. Placing an additional electron requires it to be placed in a higher energy level, which requires inputting energy into the system, making the process endothermic.
How does the electron configuration of an element influence its electron affinity?
-The electron configuration significantly influences electron affinity. Elements with half-filled or empty orbitals in their outermost shell tend to have exothermic electron affinities because adding an electron to these orbitals results in a stable configuration. Conversely, elements with filled orbitals, where an electron must be added to a higher energy level, tend to have endothermic electron affinities due to the instability caused by electron repulsion in the new orbital.
Outlines
🔬 Atomic Radius and Periodic Trends
This paragraph discusses how atomic radius varies across the periodic table. As you move left or down the table, the atomic radius increases. The size difference between hydrogen and helium is explained by the greater nuclear charge in helium, which pulls electrons closer, resulting in a smaller atomic radius. The influence of nuclear charge and electron distance on atomic size is also covered, with examples of lithium being larger than hydrogen despite having more protons.
📊 Ranking Elements by Atomic Size
The paragraph explains the concept of ranking elements by their atomic size. It uses examples of magnesium, phosphorus, and chlorine to illustrate how atomic size decreases from left to right across a period, and increases down a group. The paragraph also discusses the shielding effect of inner core electrons and how it impacts the atomic size, using chlorine, magnesium, and phosphorus as examples.
🚦 Ionic Radii and Their Comparison
This section covers the differences between atomic and ionic radii. It explains that cations, having fewer electrons, are smaller than their parent atoms, while anions, with additional electrons, are larger. The trend of ionic radii is similar to atomic radii, increasing down a group and to the left across a period. The paragraph also discusses isoelectronic species and how to rank them by size based on their atomic number.
⚛️ Electronegativity and Metallic Character
The paragraph delves into electronegativity, which is the ability of an atom to attract electrons. It increases towards the upper right of the periodic table, with fluorine being the most electronegative. Metals, located on the left, tend to be electropositive and lose electrons easily, while nonmetals, on the right, are electronegative and gain electrons. The paragraph also ranks elements such as silicon, magnesium, chlorine, and aluminum in order of increasing electronegativity.
🔋 Metallic Character and Its Variation
This part discusses metallic character, which increases as you move down a group and towards the left on the periodic table. It provides examples of elements like silicon, sodium, sulfur, aluminum, and chlorine, and ranks them in order of increasing metallic character. The paragraph explains that sodium, despite having a higher nuclear charge than aluminum, has a greater metallic character due to its willingness to lose electrons.
⚡ Ionization Energy and Its Trends
The paragraph explores ionization energy, which is the energy required to remove an electron from a gaseous atom. It tends to be lower for metals and higher for nonmetals. Ionization energy generally increases across a period from left to right and decreases down a group. Exceptions to this trend are also discussed, such as the decrease in ionization energy when moving from beryllium to boron due to the 2p orbital being higher in energy and further from the nucleus.
💥 Exceptions to Ionization Energy Trends
This section highlights exceptions to the ionization energy trends, such as the temporary decrease when moving from nitrogen to oxygen or phosphorus to sulfur. It explains that this is due to electron repulsion in the orbitals of the elements, making it easier to remove the valence electron. The paragraph also provides examples of how to rank elements by their first ionization energy and introduces the concept of higher ionization energies required for removing core electrons.
🌟 Electron Affinity and Its Trends
The paragraph discusses electron affinity, which is the energy change that occurs when an electron is added to a gaseous atom. Halogens exhibit strong exothermic electron affinity due to their high electronegativity. Trends in electron affinity are generally exothermic as you move right across a period and endothermic for groups 2 and 8. The stability of the resulting ion after electron addition is a key factor in determining whether the process is exothermic or endothermic.
📉 Electron Affinity: Endothermic vs Exothermic
This part explains the conditions that make electron affinity exothermic or endothermic. It is exothermic when an electron adds to a half-filled or empty orbital, creating a stable ion, and endothermic when an electron must be added to a higher energy level, as with group 2 and 8 elements. The paragraph concludes with a ranking of elements—chlorine, phosphorus, argon, magnesium, sodium, and silicon—based on their electron affinity values from most endothermic to most exothermic.
Mindmap
Keywords
💡Atomic Radius
💡Electronegativity
💡Ionization Energy
💡Electron Affinity
💡Metallic Character
💡Ionic Radius
💡Shielding Effect
💡Effective Nuclear Charge
💡Periodic Trends
💡Coulomb's Law
💡Isotopes
Highlights
Atomic radius increases as you move left or down the periodic table.
Helium is smaller than hydrogen due to a greater nuclear charge attracting electrons closer despite a higher atomic mass.
The force of attraction between protons and electrons increases with the number of charges and decreases with distance.
Lithium is larger than hydrogen despite having more protons because it has additional electron shells.
Shielding effect of inner core electrons reduces the force of attraction on outer valence electrons.
Electronegativity increases towards the upper right corner of the periodic table, with fluorine being the most electronegative element.
Metals tend to be electropositive and nonmetals tend to be electronegative, influencing their tendency to lose or gain electrons.
Ionization energy is the energy required to remove an electron and generally increases from left to right across a period.
Ionization energy decreases down a group due to increasing distance between the valence electrons and the nucleus.
Electron affinity is the energy change when adding an electron to a gaseous atom, with halogens showing the most exothermic reactions.
Electron affinity generally increases as you move to the right across a period, but with several exceptions.
Group 7 elements (halogens) are the most exothermic in electron affinity due to their high electronegativity.
Elements in group 2 and 8 tend to be endothermic in electron affinity because adding an electron requires energy input.
Electron affinity can be exothermic or endothermic depending on whether the added electron creates a stable or unstable ion.
The electron configuration of elements plays a crucial role in determining their electron affinity.
Electronegativity values for elements like boron, carbon, nitrogen, oxygen, fluorine, phosphorus, sulfur, chlorine, bromine, and iodine are provided.
Metallic character increases as you move down a group and from left to right across the periodic table.
The distance between the nucleus and valence electrons plays a significant role in ionization energy, often more so than nuclear charge.
Transcripts
Browse More Related Video

Periodic Trends - Atomic Radius, Electronegativity, Ionization Energy - Chemistry Series

Periodic Trends of the Periodic Table

Trends in the Periodic Table

7.5 Periodic Trends | High School Chemistry
Periodic Table Trends: Ionization Energy

Atomic radius trends on periodic table | Periodic table | Chemistry | Khan Academy
5.0 / 5 (0 votes)
Thanks for rating: