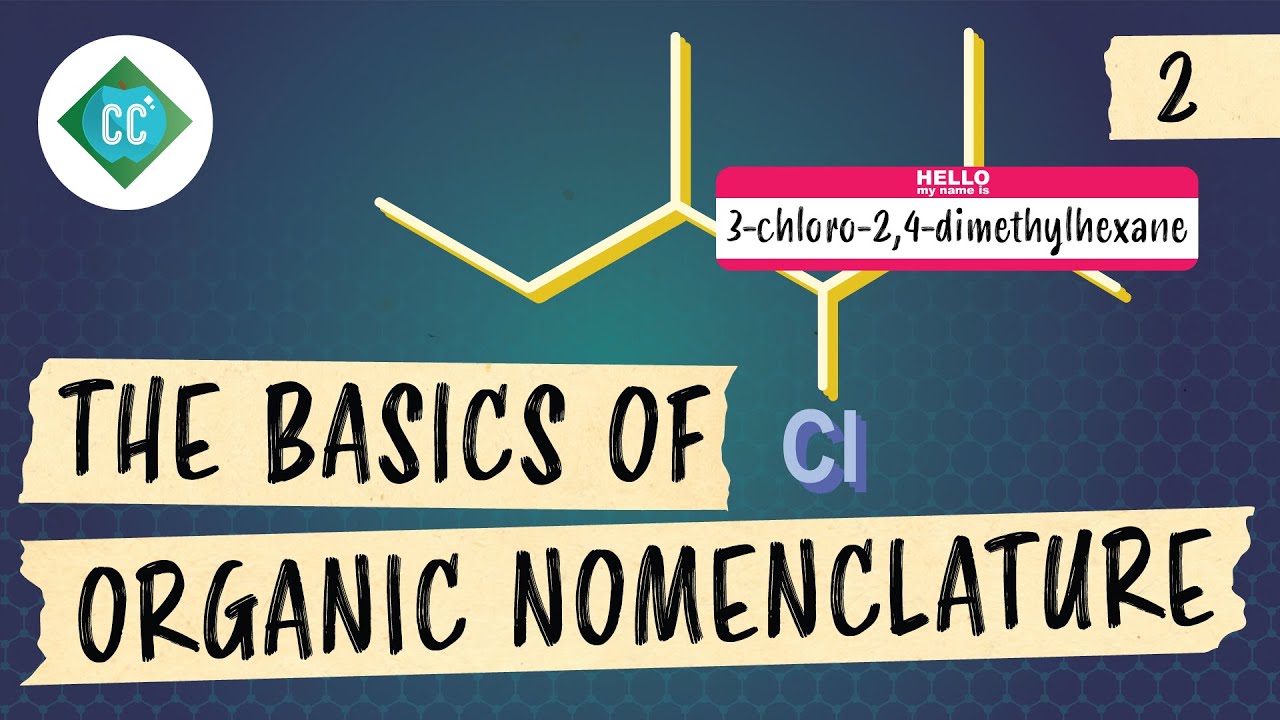
IUPAC Nomenclature of Haloalkanes and Alcohols
TLDRThis tutorial script delves into the nomenclature of hydrocarbons with various substituents, such as alkyl, halogen, and hydroxyl groups. It emphasizes the importance of identifying the longest carbon chain and numbering it strategically to prioritize certain substituents, like hydroxyl, over others. The script outlines the process of naming these compounds, including the use of prefixes for multiple substituents and the alphabetic order of substituents. It concludes with a reminder to list substituents alphabetically, disregarding most prefixes for this purpose, and invites viewers to subscribe for more educational content.
Takeaways
- 🧪 Haloalkanes are hydrocarbons with halogen substituents, such as bromine, chlorine, fluorine, or iodine.
- 🔍 When identifying the longest carbon chain, non-carbon atoms like halogens are not counted as part of the chain.
- ➡️ The direction of numbering the carbon chain can be from right to left or left to right, depending on which gives the substituent occurring soonest.
- 📝 Substituents are listed alphabetically, with prefixes indicating the frequency of their occurrence, except for 'di' which is not considered for alphabeticity.
- 🌟 The presence of a hydroxyl group changes the suffix from 'ane' to 'ol', indicating an alcohol, and it takes priority in numbering over alkyl and halogen groups.
- 🔢 The hydroxyl group's position is included in the name, and it dictates the direction of numbering to ensure it is listed first if it has the highest priority.
- 📌 The numbering of the carbon chain is separate from the listing of substituents; the two processes serve different purposes in the naming convention.
- 📚 The naming of simple hydrocarbons involves two main steps: identifying the longest carbon chain and then numbering and listing substituents alphabetically.
- ✂️ In cases where a substituent occurs on the same carbon from either direction of numbering, the direction that lists the substituent alphabetically first is chosen.
- 📝 The tutorial emphasizes the importance of following IUPAC naming rules for accurate and consistent chemical nomenclature.
- 📧 The speaker invites viewers to subscribe for more tutorials and to reach out with questions, indicating an open line of communication for further learning.
Q & A
What are haloalkanes?
-Haloalkanes are hydrocarbons with halogen substituents, such as bromine, chlorine, fluorine, or iodine.
How do you determine the longest carbon chain in a haloalkane?
-Identify the longest continuous carbon chain, ensuring not to count the halogen atoms as carbons.
How should you number the carbon chain in a haloalkane?
-Number the carbon chain from the end that gives the first substituent the lowest possible number.
What is the correct order for naming substituents alphabetically in haloalkanes?
-List substituents alphabetically, ignoring prefixes like di-, tri-, etc., except for 'iso'.
How do you name a molecule with multiple halogen substituents?
-Use prefixes (di-, tri-, etc.) to indicate the number of each type of halogen and list their positions on the carbon chain.
What is an alcohol in terms of organic chemistry?
-An alcohol is a hydrocarbon that bears a hydroxyl group (OH) attached to one of its carbon atoms.
How does the presence of a hydroxyl group affect the naming of a molecule?
-The suffix changes from -ane to -ol, and the position of the hydroxyl group must be specified.
What priority does the hydroxyl group have in numbering the carbon chain?
-The hydroxyl group takes priority over alkyl and halogen substituents, so the chain is numbered to give the hydroxyl group the lowest possible number.
Provide an example of naming a molecule with both halogen and hydroxyl groups.
-For a molecule with a hydroxyl group on carbon 2 and an iodo group on carbon 6 of a hexane chain, the name would be 6-iodo-2-hexanol.
What are the two main steps in naming a simple hydrocarbon with substituents?
-First, identify the longest carbon chain and number it to give substituents the lowest possible numbers. Second, list the substituents alphabetically, using prefixes to indicate multiple identical substituents.
Outlines
🌟 Haloalkanes and Halogen Substituents Naming
This paragraph introduces the concept of haloalkanes, which are hydrocarbons with halogen substituents. It explains the process of naming these compounds by identifying the longest carbon chain, numbering it from the direction that places the substituent earliest in the sequence, and then listing the substituents alphabetically. The paragraph emphasizes the importance of considering the presence of halogens (bromo, chloro, fluoro, iodo) and how they are abbreviated in the compound's name. It also clarifies that the prefix 'di' is not considered in alphabetical order and provides an example of naming a compound with multiple bromine atoms and other substituents.
🍶 Alcohols and Hydroxyl Group Prioritization
The second paragraph delves into the naming of alcohols, which are hydrocarbons containing a hydroxyl (OH) group. It highlights that the presence of a hydroxyl group changes the suffix of the hydrocarbon from 'ane' to 'ol'. The paragraph explains the priority of numbering the carbon chain to ensure the hydroxyl group is listed first in the name, even before alkyl or halogen substituents. An example is given to illustrate the process of naming a compound with a hydroxyl group and an iodo substituent, emphasizing the importance of alphabetic order and the frequency of substituents in the naming convention. The paragraph concludes with a summary of the steps required to name simple hydrocarbons, including identifying the longest chain and numbering it to give priority to the substituent occurring the soonest.
Mindmap
Keywords
💡Haloalkanes
💡Hydroxyl group
💡Alcohol
💡Substituents
💡Longest carbon chain
💡Numbering carbon chain
💡Alphabeticity
💡Functional group priority
💡Hydrocarbon
💡Methyl group
Highlights
Introduction to the naming of hydrocarbons with halogen substituents, referred to as haloalkanes.
Explanation of how to identify the longest carbon chain while excluding non-carbon atoms like bromine.
Direction of numbering the carbon chain based on the position of substituents.
Abbreviation of halogen groups in the naming of haloalkanes.
Alphabetical rules for substituents in naming haloalkanes.
The use of prefixes to indicate the frequency of substituents.
Introduction to alcohols and the naming convention change due to the presence of a hydroxyl group.
Suffix change from 'ane' to 'ol' for alcohols.
Prioritization of the hydroxyl group in the numbering of carbon chains.
Direction of numbering influenced by the presence of a hydroxyl group.
Naming a hydrocarbon with multiple substituents, including a hydroxyl group.
Summary of the steps for naming simple hydrocarbons.
Emphasis on the importance of identifying the longest carbon chain first.
The necessity of numbering the chain to give priority to certain substituents.
Listing of substituents alphabetically and the use of prefixes for frequency.
Instruction to disregard most prefixes for alphabeticity except 'iso'.
Invitation to subscribe for more tutorials and an offer to answer questions via email.
Transcripts
5.0 / 5 (0 votes)
Thanks for rating: