Carbanion Stability
TLDRThis video delves into the stability of carbon ions, contrasting the stability of carbocations and carbanions. It explains that tertiary carbocations are the most stable due to hyperconjugation and inductive effects from electron-donating methyl groups. Conversely, carbanions are more stable when they are primary, as electron-donating groups destabilize them. The video also highlights that allylic carbanions are stabilized through resonance, spreading the negative charge over multiple carbon atoms, making them more stable than those with localized charges.
Takeaways
- 🔬 A carbocation is a carbon atom with a positive charge.
- 🔄 Carbocations are classified as primary, secondary, or tertiary based on the number of carbon atoms they are attached to.
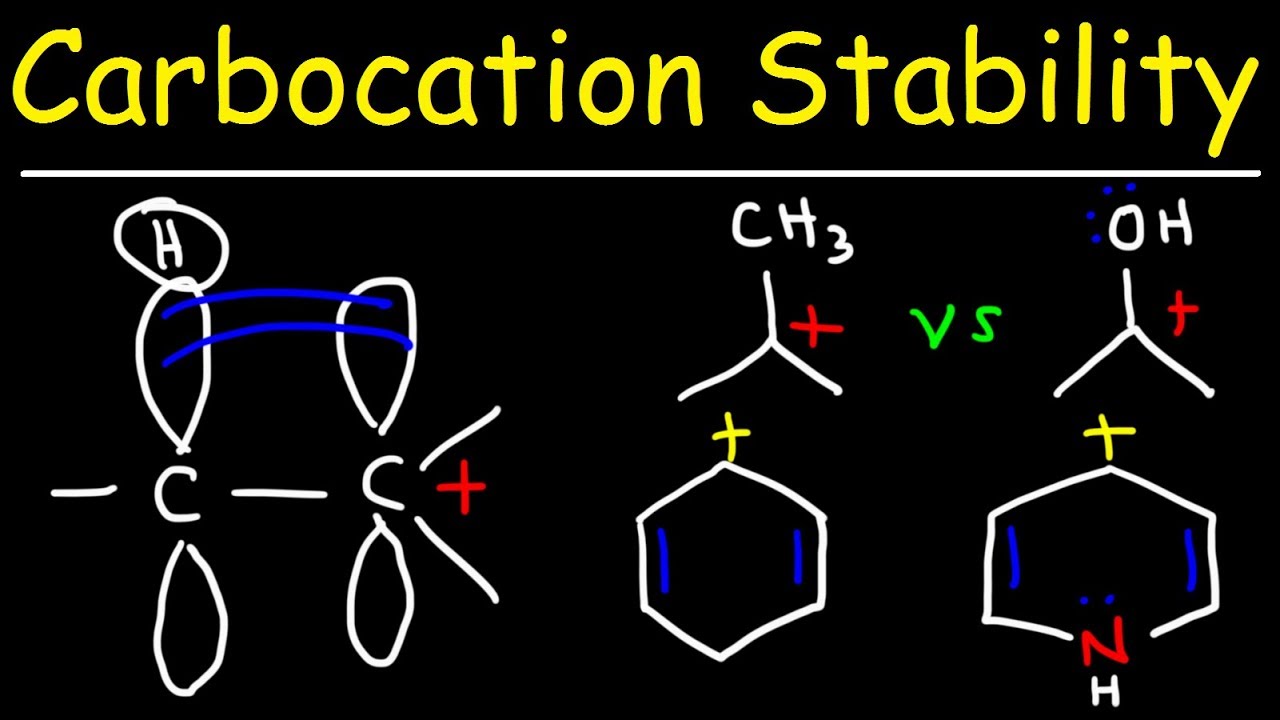
- 🧲 Tertiary carbocations are more stable than secondary and primary carbocations due to hyperconjugation and inductive effects.
- 🌐 Hyperconjugation involves orbital overlap between a nearby C-H bond, stabilizing the carbocation.
- 🍃 The inductive effect is the stabilization of a carbocation by electron-donating groups like methyl groups.
- 📉 Methyl groups increase the stability of carbocations, with tertiary carbocations being the most stabilized.
- ⚠️ In contrast to carbocations, electron-donating groups destabilize carbanions due to their negative charge.
- 🔁 Carbanions are more stable when the negative charge is delocalized over multiple atoms, as seen in allylic carbanions.
- 🔄 Resonance structures contribute to the stability of ions by spreading the charge over multiple atoms.
- 📚 Understanding the stability of carbocations and carbanions is crucial for organic chemistry exams.
- 👋 The video provides insights into carbocation and carbanion stability, aiming to assist with organic chemistry studies.
Q & A
What is a carbocation?
-A carbocation is a carbon atom with a positive charge, typically bonded to fewer hydrogen atoms than usual, resulting in an overall positive charge.
Why are tertiary carbocations more stable than secondary or primary carbocations?
-Tertiary carbocations are more stable due to two main reasons: the hyperconjugation effect and the inductive effect, which both contribute to the stabilization of the positive charge.
What is the hyperconjugation effect and how does it stabilize a carbocation?
-The hyperconjugation effect is the interaction between the filled σ-bond of an adjacent carbon atom and the empty p-orbital of the positively charged carbon atom, which helps to stabilize the carbocation.
What is the inductive effect and how does it relate to carbocation stability?
-The inductive effect is the stabilization of a carbocation by electron-donating groups such as methyl groups, which can donate electron density through sigma bonds to the positively charged carbon.
Why are carbanions less stable when electron-donating groups are present?
-Carbanions, which have a negative charge, are less stable when electron-donating groups are present because the additional electron density makes the anion more electron-rich, thus less stable.
How does the stability of carbon ions differ between carbocations and carbanions?
-Carbocations are more stable with more electron-donating groups, making tertiary carbocations the most stable. Conversely, carbanions are more stable with fewer electron-donating groups, making tertiary carbanions the least stable.
What is an allylic carbocation and why is it more stable than a regular secondary carbocation?
-An allylic carbocation is a secondary carbocation that is part of an allylic system, which allows for charge delocalization through resonance, making it more stable than a regular secondary carbocation.
How does resonance contribute to the stability of allylic carbocations?
-Resonance allows the negative charge in an allylic carbocation to be delocalized over two carbon atoms instead of one, leading to a more stable configuration.
What happens when you move a lone pair to form a pi bond in an allylic carbocation?
-Moving a lone pair to form a pi bond in an allylic carbocation leads to the creation of a resonance structure where the negative charge is shared between two carbon atoms, increasing stability.
How does the concept of charge delocalization relate to the stability of carbon ions?
-Charge delocalization over multiple atoms in carbon ions, as seen in resonance structures, results in a more stable configuration compared to when the charge is localized on a single atom.
What advice does the video offer for students preparing for organic chemistry exams?
-The video suggests understanding the concepts of carbocation and carbanion stability, including the effects of hyperconjugation, inductive effects, and resonance, to help with organic chemistry exams.
Outlines
🔬 Carbocation Stability and Inductive Effects
This paragraph delves into the concept of carbocations, which are carbon atoms with a positive charge. It explains the stability hierarchy of carbocations, with tertiary being more stable than secondary and primary due to hyperconjugation and inductive effects. The inductive effect is illustrated through the electron-donating nature of methyl groups, which stabilize carbocations by donating electron density through sigma bonds. Hyperconjugation is described as the orbital overlap between a nearby C-H bond and the carbocation, further stabilizing it. The paragraph also contrasts carbocations with carbon anions, noting that electron-donating groups have a different effect on stability for negatively charged ions.
🌐 Resonance and Stability in Carbon Ions
The second paragraph contrasts the stability of carbon ions, focusing on the impact of resonance structures. It introduces the concept of allylic carbon ions, which are more stable due to the delocalization of the negative charge across multiple carbon atoms. The explanation includes the process of drawing resonance structures, demonstrating how the negative charge can be spread over two carbon atoms, making the situation more stable than if the charge were localized on a single atom. This paragraph emphasizes the importance of resonance structures in determining the stability of carbon ions in organic chemistry.
Mindmap
Keywords
💡Carbo cation
💡Tertiary carbo cation
💡Secondary carbo cation
💡Hyperconjugation effect
💡Inductive effect
💡Methyl group
💡Carbon anion
💡Allylic carbon
💡Resonance
💡Pi bond
💡Stability
Highlights
A carbo cation is a carbon with a positive charge.
Tertiary carbo cations are more stable than secondary or primary carbo cations.
Stability of carbo cations is influenced by hyperconjugation and inductive effects.
Methyl groups are electron-donating groups that stabilize carbo cations through the inductive effect.
Hyperconjugation effect involves orbital overlap between a nearby C-H bond and the carbo cation.
Tertiary carbo cations are more stable due to the presence of more methyl groups.
Carbon anions differ in stability; methyl carbon anions are more stable than tertiary carbon anions.
Electron-donating groups destabilize carbon anions as they already have a negative charge.
Secondary allylic carbo cations are more stable due to resonance, allowing charge delocalization.
Resonance structures spread the negative charge over multiple atoms, increasing stability.
Comparing carbon ions, those with resonance structures are generally more stable.
The video aims to aid understanding of carbon ion stability for organic chemistry exams.
Carbon ion stability is crucial for grasping organic chemistry concepts.
Understanding the differences in stability between various types of carbon ions is key.
The inductive effect and hyperconjugation are fundamental to carbo cation stability.
Methyl groups play a significant role in the stabilization of tertiary carbo cations.
Resonance stabilization is a critical concept in organic chemistry for understanding carbon ion stability.
The video provides a comprehensive overview of carbon ion stability for educational purposes.
Transcripts
Browse More Related Video
Carbocation Stability - Hyperconjugation, Inductive Effect & Resonance Structures

6.4 Nucleophiles, Electrophiles, and Intermediates | Organic Chemistry
14.6a Fragmentation Patterns of Alkanes, Alkenes, and Aromatic Compounds | Organic Chemistry

[H2 Chemistry] 2021 Topic 11 Alkenes In-Class Exercise 1 Review

Resonance Structures

Carbenes Part 1: Properties and Formation
5.0 / 5 (0 votes)
Thanks for rating: