Periodic Table of Elements Explained - Metals, Nonmetals, Valence Electrons, Charges
TLDRThis educational video script delves into the intricacies of the periodic table, highlighting the properties and characteristics of various elements. It discusses the reactivity of alkali metals, the formation of cations and anions, and the distinct behaviors of metals and nonmetals. The script also explores the roles of valence electrons in determining an element's chemical behavior, introduces the concept of metalloids, and explains the significance of atomic numbers and mass in identifying elements. Furthermore, it touches on the physical states of elements, the conductivity of metals, and the inert nature of noble gases, providing a comprehensive foundation for understanding the periodic table.
Takeaways
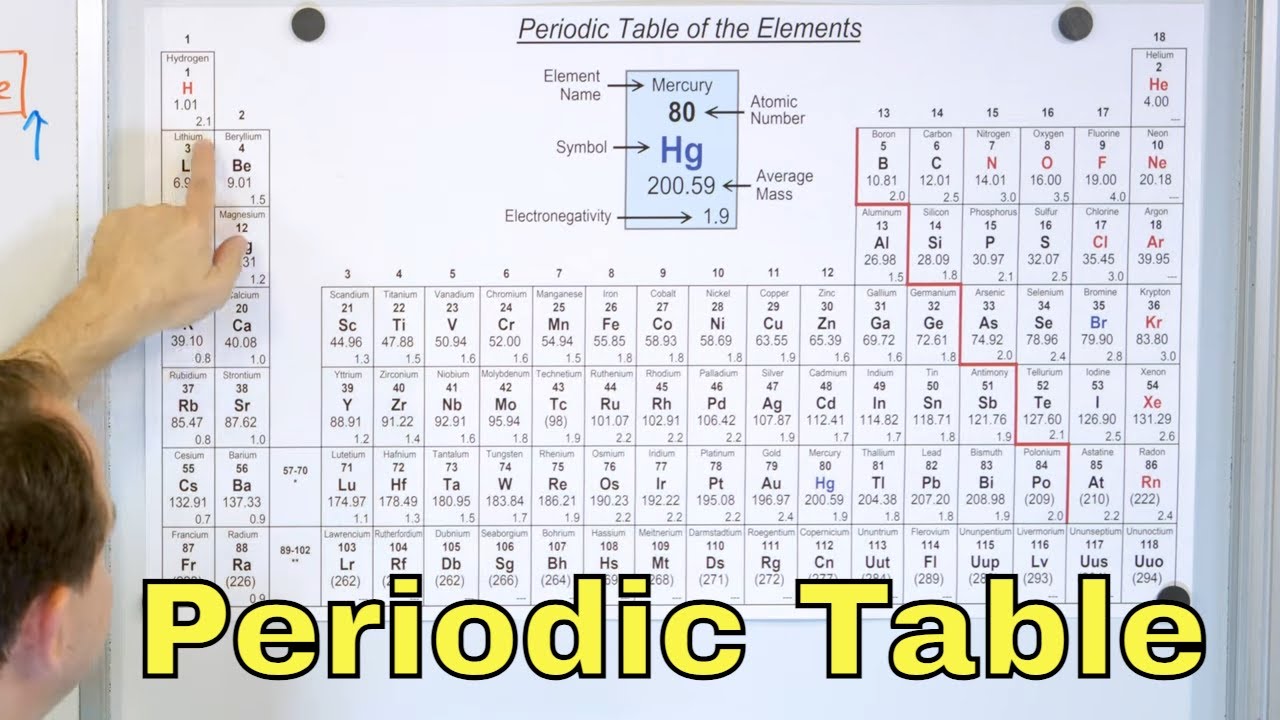
- 🔬 The periodic table is organized by elements' properties, with the first column including hydrogen and alkali metals like lithium, sodium, and potassium, which are reactive and tend to form cations with a +1 charge.
- 🌟 Hydrogen is unique as a nonmetal in the first column, while the rest are metals. Metals are known for their reactivity and ability to give away electrons.
- 📊 Group 2 elements, the alkaline earth metals, have two valence electrons and form ions with a +2 charge, and are less reactive compared to alkali metals.
- 🛑 Transition metals, found in the middle of the periodic table, vary in reactivity and include common elements like zinc, copper, iron, silver, gold, platinum, and mercury.
- ⚛️ Group 8, the noble gases, are chemically inert with helium having two valence electrons and others like neon having eight, making them stable and unreactive.
- 💥 Halogens, including fluorine, chlorine, bromine, and iodine, are very reactive nonmetals with seven valence electrons, forming negative 1 charges as ions.
- 🔩 Chalcogens (Group 6a) have six valence electrons and tend to form negative 2 charges, while elements with five valence electrons like nitrogen, phosphorus, and arsenic form negative 3 charges.
- 🔩 Metals are found on the far left of the periodic table and are characterized by their ability to conduct heat and electricity, being malleable and ductile.
- ⚡ Nonmetals, located on the upper right of the periodic table, do not conduct electricity or heat and are insulators, acquiring electrons to form negative charges, with fluorine being the most electronegative.
- 🌐 Metalloids, such as silicon and germanium, are situated near the metalloid line on the periodic table and exhibit properties between metals and nonmetals, conducting a small amount of electricity.
- 🔢 The atomic number represents the number of protons and is equal to the number of electrons in a neutral atom, while the atomic mass roughly equals the sum of protons and neutrons.
Q & A
What are alkali metals and how do they react with water?
-Alkali metals are elements found in Group 1 of the periodic table, including lithium, sodium, and potassium. They are very reactive and when placed in water, they tend to explode due to their eagerness to give away their valence electrons.
How many valence electrons do alkaline earth metals have and what ions do they form?
-Alkaline earth metals, found in Group 2 of the periodic table, have two valence electrons. They form ions with a +2 charge.
What are some common symbols for transition metals and their general reactivity?
-Some common symbols for transition metals include Zn for zinc, Cu for copper, Fe for iron, Ag for silver, Au for gold, Pt for platinum, and Hg for mercury. Their reactivity varies, with some being reactive and others not.
What is the general characteristic of noble gases in terms of reactivity?
-Noble gases, found in Group 18, are chemically inert and do not readily react with other elements due to their stable electron configurations, typically having a full outer shell of electrons.
How many valence electrons do halogens have and what ions do they form?
-Halogens, found in Group 17, have seven valence electrons and they form negative 1 charges as ions due to their tendency to gain one electron to achieve a full outer shell.
What is the difference between metals and nonmetals in terms of electron affinity?
-Metals tend to give away electrons and form positive charges (cations), making them electropositive. Nonmetals, on the other hand, tend to acquire electrons and form negative charges (anions), making them electronegative.
What are metalloids and how do their electrical conductivity compare to metals and nonmetals?
-Metalloids are elements that have properties intermediate between metals and nonmetals. They are not pure insulators like nonmetals and do not conduct electricity as well as metals. However, they do conduct a small amount of electricity and their conductivity can increase with light exposure or temperature.
What does the atomic number represent and how is it related to the number of protons in an element?
-The atomic number represents the number of protons in the nucleus of an atom and is equal to the number of electrons in a neutral atom. It is also the defining characteristic of an element.
How can you determine the number of neutrons in an atom?
-The number of neutrons in an atom can be determined by subtracting the atomic number (number of protons) from the mass number (total number of protons and neutrons).
What is the significance of the strong nuclear force in the nucleus of an atom?
-The strong nuclear force is what keeps the protons and neutrons together in the nucleus of an atom. It counteracts the electrostatic repulsion that would otherwise cause the positively charged protons to repel each other and move apart.
What are the physical states of the diatomic elements at room temperature?
-Diatomic elements such as hydrogen, nitrogen, oxygen, fluorine, chlorine, and iodine exist as gases at room temperature. Bromine is a red liquid, and the physical state of other elements like gallium can change with temperature, as it can melt into a liquid with slight heating.
Outlines
🔬 Introduction to the Periodic Table
This video discusses the periodic table and properties of elements. It begins with Group 1 elements, such as hydrogen (a nonmetal), lithium, sodium, and potassium (alkali metals). These elements are highly reactive and have one valence electron, typically forming a +1 charge as ions. Group 2 elements, including beryllium, magnesium, and calcium, are alkaline earth metals with two valence electrons and a +2 charge. Transition metals like zinc, copper, and iron are also mentioned, noting their varied reactivity and common symbols.
🧪 Characteristics of Various Element Groups
The video continues by describing other element groups such as boron, carbon, nitrogen, oxygen, and noble gases like helium and neon, which are chemically inert. Halogens (fluorine, chlorine, bromine, iodine) have seven valence electrons and form -1 charges, while chalcogens (oxygen, sulfur, selenium) have six valence electrons and form -2 charges. The periodic table's structure and the properties of metals, nonmetals, and metalloids are also explained.
⚛️ Atomic Structure and Electron Configuration
This section covers the atomic structure, focusing on the nucleus (protons and neutrons) and electron configuration. The concept of valence electrons and core electrons is explained using lithium as an example. The interaction of electric forces within the atom and the role of the strong nuclear force in maintaining atomic stability are discussed.
🧮 Calculating Atomic Particles
The video delves into finding the number of electrons, protons, and neutrons in an atom, using aluminum as an example. The process of determining these particles for atoms and ions is detailed, along with a brief explanation of isotopes. The importance of the atomic number and mass number is highlighted.
🔍 Element Identification and Quizzes
This part of the video provides quizzes to help viewers identify elements by their symbols and properties. It includes exercises to name elements based on their symbols and to determine the number of valence electrons, charges, and whether elements are metals or nonmetals.
🧲 Element Reactivity and Conductivity
The discussion covers which elements are likely to form negatively charged ions (anions) and which are metals, nonmetals, or metalloids. The reactivity of alkali metals, alkaline earth metals, transition metals, and inner transition metals is analyzed, along with the concept of chemical inertness and electrical conductivity of elements like carbon (in the form of graphite) and nickel.
🌡️ States of Matter and Special Elements
The final section explains the physical states of elements at room temperature, focusing on diatomic elements and exceptions like mercury (a liquid metal) and gallium (which melts near room temperature). The video concludes by summarizing key points about the periodic table and element properties.
Mindmap
Keywords
💡Periodic Table
💡Alkali Metals
💡Valence Electrons
💡Alkaline Earth Metals
💡Transition Metals
💡Noble Gases
💡Halogens
💡Metalloids
💡Electronegativity
💡Atomic Number
💡Ions
Highlights
Introduction to the periodic table and properties of elements.
Explanation of Group 1 elements, including hydrogen and alkali metals, and their reactivity.
Details on alkali metals' tendency to form positively charged ions (cations) with a +1 charge.
Description of Group 2 elements, the alkaline earth metals, and their formation of +2 charged ions.
Overview of transition metals, their reactivity, and common elements like zinc, copper, iron, and gold.
Introduction to noble gases, their chemical inertness, and their electron configurations.
Discussion on halogens as highly reactive nonmetals with seven valence electrons and their tendency to form negative ions.
Explanation of chalcogens, Group 6a elements, and their typical formation of negative 2 charges.
Information on elements with five valence electrons, such as nitrogen and phosphorus, and their common -3 charges.
Description of metalloids, their properties, and examples like silicon and germanium.
Differentiation between metals, nonmetals, and metalloids based on their electrical conductivity and electron affinity.
Identification of the most electronegative element, fluorine, and the most electropositive metal, francium.
Explanation of atomic structure, including the nucleus with protons and neutrons, and electron orbits.
Clarification of the strong nuclear force that holds the nucleus together despite the repulsive forces between protons.
Tutorial on calculating the number of protons, neutrons, and electrons in an atom or ion.
Quizzes to test knowledge on element identification, properties, and classifications.
Differentiation between metals, nonmetals, and metalloids in terms of conductivity and chemical reactivity.
Identification of elements by their symbols and their physical states at room temperature.
Conclusion summarizing the importance of understanding the periodic table and element properties.
Transcripts
Browse More Related Video

Periodic Table of Elements - Element Classes

Periodic Table of Elements Song
The Periodic Table of the Elements in Chemistry - [1-2-12]

Periodic Table Explained: Name Origin

The periodic table | Atoms, elements, and the periodic table | High school chemistry | Khan Academy

3.2 Introduction to the Periodic Table | High School Chemistry
5.0 / 5 (0 votes)
Thanks for rating: