The MOLE & Avogadro's Number (Chemistry)
TLDRThe video script delves into the concept of counting large quantities of objects, particularly in chemistry, using specific terms like 'pair,' 'dozen,' and 'mole.' It explains that a mole represents 6.02 x 10^23 of a substance's representative particles, such as atoms, ions, or molecules. The script introduces Avogadro's Number, which is the basis for the mole, and its significance in chemistry due to the minuscule size of particles. The video also demonstrates how to use the mole for conversions between mass and the number of particles, especially for gases at standard temperature and pressure (STP). It provides examples of calculating the number of water molecules in a beaker and the mass and number of atoms in a neon sign, illustrating the utility of the mole in chemical calculations.
Takeaways
- 🧮 Counting units like 'pair', 'dozen', and 'ream' are used to simplify the counting of specific items.
- 🥚 'Pair' refers to two items, 'dozen' is a set of 12, and 'ream' is a package of 500 sheets of paper.
- 🔍 A 'mole' is a counting unit used in chemistry to represent a very large number of objects, specifically 6.02 x 10^23.
- ⚛️ The mole is used to count atoms, ions, formula units, or molecules, which are the smallest units of a material.
- 🎓 Avogadro's number, 6.02 x 10^23, is named after the Italian chemist Amedeo Avogadro and is central to chemistry for counting particles.
- ⚖️ The molar mass of a substance is the mass of one mole of that substance and is used to convert between mass and moles.
- 💧 For water, the molar mass is 18.015 g/mol, calculated by adding the atomic masses of its constituent hydrogen and oxygen atoms.
- 🧪 To find the number of molecules in a sample, use the molar mass and Avogadro's number as conversion factors.
- 🌡️ At standard temperature and pressure (STP), 1 mole of any gas occupies 22.4 liters.
- 🤔 For gases, conversions between volume and number of particles can be made using the volume at STP and Avogadro's number.
- 📏 To convert between different units (mass, moles, number of particles, volume for gases), use the appropriate conversion factors like molar mass and Avogadro's number.
- 📚 Understanding and applying the concept of the mole is essential for solving various chemical conversion problems.
Q & A
What is the significance of the term 'pair' when counting certain objects?
-The term 'pair' is used to refer to two items that typically come together, such as shoes and socks, making counting easier by referring to them in pairs.
What does the term 'dozen' represent in terms of quantity?
-A 'dozen' represents a quantity of 12 items, commonly used when buying items like eggs.
What is a 'ream' of paper and how many sheets does it contain?
-A 'ream' of paper refers to a specific quantity of paper, which is 500 sheets.
What is the term used to count a very large number of objects in chemistry?
-The term 'mole' is used to count a very large number of objects, specifically 6.02 x 10^23 objects, in chemistry.
What are the different types of particles that a mole can represent in chemistry?
-A mole can represent atoms, ions, formula units in the case of ionic compounds, or molecules in the case of molecular compounds.
Who is Avogadro's number named after and what is its approximate value?
-Avogadro's number is named after the Italian chemist Amedeo Avogadro and its approximate value is 6.02 x 10^23.
Why is the number 6.0221421 x 10^23 significant in chemistry?
-The number 6.0221421 x 10^23 is significant because it represents the number of atoms in 12 grams of Carbon-12, which is used as a standard in chemistry.
How can you convert grams of water to the number of moles of water?
-To convert grams of water to moles, you use the molar mass of water, which is the mass of one mole of water, and divide the mass of water in grams by the molar mass.
What is the molar mass of water and how is it calculated?
-The molar mass of water is approximately 18.0 g/mol. It is calculated by adding the atomic masses of the two hydrogen atoms and one oxygen atom in a water molecule from the Periodic Table.
How many molecules of water are in 400.0 grams of water?
-In 400.0 grams of water, there are 1.3 x 10^25 molecules of water, calculated using the molar mass of water and Avogadro's number.
What is the relationship between the volume of a gas and the number of moles at standard temperature and pressure (STP)?
-At STP, 1 mole of any gas occupies 22.4 liters. This relationship allows for the conversion between the volume of a gas and the number of moles.
How many atoms of neon are in 200 mL of neon gas at standard temperature and pressure (STP), and how many grams does it weigh?
-In 200 mL of neon gas at STP, there are 5.4 x 10^21 atoms of neon, and it weighs approximately 0.18 grams.
Outlines
🧬 Understanding Counting Units in Chemistry
This paragraph introduces the concept of counting units like pairs, dozens, and reams, which are used to simplify counting in everyday situations. It then transitions into the use of the term 'mole' in chemistry to count a very large number of objects, specifically atoms, ions, formula units, or molecules. The mole is defined as 6.02 x 10^23 representative particles and is named after the Italian chemist Amedeo Avogadro. The paragraph further explains the application of the mole in calculating the number of particles in a given mass of a substance, using water as an example. It also touches on the molar mass concept and how it is used to convert between grams and moles, and then to the number of molecules using Avogadro's number.
📏 Conversions Using Moles: Mass, Volume, and Particles
The second paragraph delves into the process of converting between grams of a substance, such as water, to the number of molecules it contains using the mole as a fundamental unit. It outlines the calculation steps, starting from the known mass of water, through the use of molar mass and Avogadro's number, to arrive at the total number of molecules. The paragraph also discusses the unique property of gases at standard temperature and pressure (STP), where 1 mole of any gas occupies 22.4 liters. An example is provided to calculate the number of neon atoms in a neon sign and its corresponding mass, using the volume of neon gas, the molar volume at STP, and the molar mass of neon. The paragraph concludes with a summary of the conversion relationships that involve the mole, molar mass, and Avogadro's number.
🔑 The Mole: Central to Chemical Conversions
The final paragraph emphasizes the pivotal role of the mole in performing various conversion problems in chemistry. It succinctly states that the mole is essential for converting between mass and moles, moles and the number of particles, and for gases, between volume and moles at STP. The paragraph reinforces the importance of understanding the mole as a key concept for chemical calculations and conversions.
Mindmap
Keywords
💡Pair
💡Dozen
💡Ream
💡Mole
💡Avogadro's Number
💡Representative Particles
💡Molar Mass
💡Standard Temperature and Pressure (STP)
💡Conversion Factors
💡Periodic Table
💡Ionic Compound
Highlights
Counting words like 'pair', 'dozen', and 'ream' are used to simplify counting in specific situations.
A 'pair' refers to a set of two items, such as shoes or socks.
A 'dozen' is a unit of measurement equal to 12, commonly used for eggs.
A 'ream' of paper consists of 500 sheets.
In chemistry, a 'mole' is used to count a very large number of objects, specifically 6.02 x 10^23.
A mole represents atoms, ions, formula units, or molecules, which are the smallest units of a material.
1 mol of helium equals 6.02 x 10^23 atoms because helium is atomic.
Hydrogen, being molecular, has 1 mol representing 6.02 x 10^23 molecules of H2.
1 mol of NaCl signifies 6.02 x 10^23 formula units, as NaCl is an ionic compound.
Avogadro's Number, 6.02 x 10^23, is named after the Italian chemist Amedeo Avogadro, though he did not determine the value.
The number 6.0221421 x 10^23 is derived from the number of atoms in 12 grams of Carbon-12, a standard in chemistry.
Avogadro's number is rounded to 6.02 x 10^23 for practical use in chemistry.
The mole is used to convert grams of a substance to the number of moles using the molar mass.
The molar mass of water is 18.015 g/mol, calculated by summing the atomic masses of hydrogen and oxygen.
1 mole of water is equivalent to 6.02 x 10^23 water molecules.
For solids and liquids, the mole is commonly used to convert between mass and the number of particles.
For gases at STP (Standard Temperature and Pressure), 1 mole of any gas occupies 22.4 liters.
An example calculation shows that a neon sign holding 200 mL of neon gas contains 5.4 x 10^21 atoms of neon.
The mass of neon in the example can be calculated using the molar mass and Avogadro's number, resulting in 0.18 grams.
A conversion map is provided to relate different types of conversions using the mole concept.
Transcripts
Browse More Related Video

Introduction to Moles
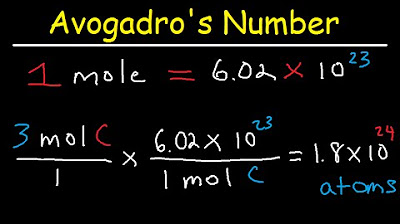
Avogadro's Number, The Mole, Grams, Atoms, Molar Mass Calculations - Introduction

Introduction to Moles

How big is a mole? (Not the animal, the other one.) - Daniel Dulek

What is a Mole? | #Extraclass #MoleConcept #Chemistry #Animation

02 - What is Avogadro's Number & the Mole in Chemistry? Part 1
5.0 / 5 (0 votes)
Thanks for rating: