How to Convert Units in Chemistry
TLDRThe video script focuses on the critical concept of unit conversion in chemistry, which is essential for success in the subject. It uses an analogy of language translation to explain the process of converting units through a chain of conversion factors. The script covers both the metric system and dimensional analysis, illustrating how to convert between units that share a common base and those that do not. It emphasizes the importance of aligning units to cancel them out and the use of scientific notation. The video provides a step-by-step guide on converting units like kilograms to grams and kilometers to centimeters, using a trick to avoid confusion between multiplication and division. It also introduces common conversion factors for dimensional analysis and explains how to apply them. The script then delves into stoichiometry, discussing how to convert between grams and moles, and moles to atoms or molecules, using molar mass and Avogadro's number. It concludes by stressing the importance of practice in unit conversion for mastering chemistry.
Takeaways
- 📚 **Unit Conversion Fundamentals**: Chemistry often involves converting units, which is crucial for understanding and success in the subject.
- 🗣️ **Language Translation Analogy**: Just as translating phrases involves using a chain of languages, unit conversion uses a chain of conversion factors.
- 🔄 **Conversion Factors**: These have two different units and help in transitioning from one unit to another, with the same units cancelling out.
- 📐 **Metric System**: Allows conversion between units that share a common base, such as grams, meters, and liters.
- 📏 **Dimensional Analysis**: Enables conversion between units that do not share a common base, like feet to inches or kilograms to pounds.
- 🤔 **Common Student Mistake**: Students often struggle with knowing whether to multiply or divide, which stems from not understanding conversion factors.
- 🔢 **Scientific Notation**: Conversion factors are often represented in scientific notation, with small and large numbers written without the leading one.
- 📉 **Base Unit Placement**: In the metric system, place the base unit with the prefix (like kilo for kilograms) on top and a '1' with the different unit on the bottom.
- ⛓ **Connecting Units**: For conversions requiring more than one factor, like kilometers to centimeters, use a common unit (meters) to connect the conversion.
- 🧠 **Memorization in Dimensional Analysis**: Students are expected to remember certain conversion factors, while others may be provided in the question.
- 🔍 **Stoichiometry**: Involves unit conversion related to elements or chemical reactions, using different conversion factors that must be known or memorized.
- ⚖️ **Molar Mass**: Allows conversion between grams and moles by using the masses from the periodic table for elements and compounds.
- 🔗 **Mole to Mole Ratios**: Used to convert moles of one compound to another, found in balanced chemical equations or through the subscripts in chemical formulas.
- 🏷️ **Avogadro's Number**: Facilitates conversion from moles to atoms, molecules, or particles, always associated with moles.
- 📊 **Standard Temperature and Pressure (STP)**: A specific conversion factor for gases that allows conversion between moles and liters at STP.
- 📈 **Significant Figures**: When rounding results, use the original numbers given in the question, not the conversion factors, to determine significant figures.
Q & A
What is the concept of unit conversion in chemistry?
-Unit conversion in chemistry is the process of changing a given value from one unit of measure to another. It is analogous to translating a phrase from one language to another through a chain of people who speak the intermediate languages.
How do conversion factors help in unit conversion?
-Conversion factors have two different units and allow us to go from one unit to a different unit. They are used to cancel out the units we want to convert from, leaving us with the desired units.
Why is it important to align the same units across from each other in unit conversion?
-Aligning the same units across from each other allows them to cancel out, which is crucial for changing the unit of the given value without altering its magnitude.
What is the role of the metric system in unit conversion?
-The metric system allows for the conversion of units that share a common base, such as going from milliliters to liters or from kilograms to milligrams. It uses prefixes like kilo-, milli-, and centi- to relate different units of the same base.
How does dimensional analysis differ from the metric system in terms of unit conversion?
-Dimensional analysis allows for the conversion of units that do not share a common base, such as going from feet to inches or kilograms to pounds. It involves using set conversion factors that are either memorized or provided in the question.
What is a common mistake students make with the metric system?
-A common mistake is not knowing whether to divide or multiply when converting units. This confusion arises from not understanding how to find and apply the correct conversion factor.
How can you determine whether to multiply or divide by a conversion factor?
-You should always place the conversion factor with the base unit on the same side as the given unit and the different unit or unit with the prefix on the opposite side. This ensures that when you multiply, the units you are converting from will cancel out.
What is the trick for setting up conversion factors in the metric system?
-The trick is to always place the number raised to an exponent (representing the metric prefix) with the base unit on top and a 1 with the different unit or unit with the prefix on the bottom.
How do you convert units that do not have a direct conversion factor, like kilometers to centimeters?
-You need to use a connecting unit that can link the two units you want to convert between. In the case of kilometers to centimeters, you would first convert kilometers to meters (a common base) and then from meters to centimeters.
What are some common conversion factors used in dimensional analysis?
-Common conversion factors include those for converting between different units of length, mass, and volume, such as feet to inches or pounds to kilograms. These factors are either memorized or provided in the question.
How does stoichiometry relate to unit conversion?
-Stoichiometry is a form of unit conversion that deals with the relationships between the amounts of reactants and products in chemical reactions. It uses conversion factors to go from grams of an element to atoms of that element or between different elements and compounds.
What conversion factors are essential to know for stoichiometry problems?
-For stoichiometry, you need to know how to calculate molar mass, understand mole to mole ratios from balanced chemical equations or chemical formulas, and be familiar with Avogadro's number for converting between moles and atoms or molecules. Additionally, the conversion factor for gases at STP (standard temperature and pressure) is important for converting between moles and liters.
Outlines
🔍 Understanding Unit Conversion and Conversion Factors
This paragraph introduces the fundamental concept of unit conversion, which is essential for success in a chemistry class. It uses the analogy of translating a phrase through a chain of languages to explain how conversion factors work. The paragraph emphasizes the importance of aligning units to cancel out and change the unit. It also distinguishes between the metric system, where units share a common base, and dimensional analysis, which deals with units that do not share a common base. The metric system allows conversion between units like milliliters to liters and kilograms to milligrams, while dimensional analysis enables conversions like feet to inches or kilograms to pounds. A common mistake is addressed, where students struggle to know whether to multiply or divide, and a trick is shared to place conversion factors correctly. Examples are provided to illustrate the process, including converting kilograms to grams and kilometers to centimeters.
📏 Mastering Dimensional Analysis and Stoichiometry
The second paragraph delves into dimensional analysis, which involves memorizing certain conversion factors and using others provided in the question. Common conversion factors are listed, and an example is given to demonstrate the process of converting feet to inches and then to centimeters. The importance of rounding to the correct number of significant figures is highlighted. The paragraph then transitions to stoichiometry, which is another form of unit conversion related to elements or chemical reactions. Molar mass and mole to mole ratios are introduced as key conversion factors in stoichiometry. Molar mass is found using the periodic table, and mole to mole ratios are derived from balanced chemical equations or chemical formulas. The paragraph also covers the memorization of Avogadro's number and the molar volume of gases at STP, which are used to convert between moles and other quantities. An example problem is presented to find the number of molecules of C2H4 from a given mass, illustrating the use of molar mass and Avogadro's number as conversion factors.
📚 Practice Makes Perfect in Chemistry
The final paragraph emphasizes the importance of practicing unit conversion and stoichiometry to pass a chemistry class. It encourages students to use the provided resources, such as practice problems, to improve their understanding and proficiency. The paragraph concludes with a call to action, inviting students to try out the practice problems and then return to the next video for further learning.
Mindmap
Keywords
💡Unit Conversion
💡Conversion Factors
💡Metric System
💡Dimensional Analysis
💡Scientific Notation
💡Molar Mass
💡Mole to Mole Ratio
💡Avogadro's Number
💡Stoichiometry
💡Significant Figures
💡Standard Temperature and Pressure (STP)
Highlights
Unit conversion is crucial for success in Chemistry class.
A conversion factor has two different units and allows transitioning from one to another.
Aligning the same units across from each other helps them cancel out, facilitating unit change.
The metric system and dimensional analysis are key areas where unit conversion is applied.
The metric system converts units sharing a common base, such as from milliliters to liters.
Dimensional analysis converts units without a common base, like feet to inches.
Students often struggle with knowing whether to multiply or divide in the metric system.
Scientific notation is used to represent small and large numbers in conversion factors.
A trick for unit conversion: Place the base unit with the prefix on top and a '1' with the different unit on the bottom.
Multiple conversion factors are used when there's no direct conversion available, like kilometers to centimeters.
Dimensional analysis requires memorizing set conversion factors and using them in calculations.
Common conversion factors include those for length, mass, volume, and time.
Stoichiometry involves unit conversion related to elements or chemical reactions.
Molar mass is used to convert between grams and moles, and vice versa.
Mole to mole ratios are found in balanced chemical equations or through the subscripts in chemical formulas.
Avogadro's number is used to convert from moles to atoms, molecules, or particles.
At STP, the volume conversion factor allows converting from moles to liters for gases.
When converting grams to molecules, use molar mass as the first conversion factor and Avogadro's number as the second.
Practicing unit conversion is essential for passing Chemistry class.
Transcripts
Browse More Related Video

Know This For Your Chemistry Final Exam - Stoichiometry Review

How to Determine Your Units in Chemistry
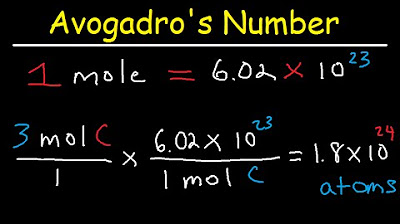
Avogadro's Number, The Mole, Grams, Atoms, Molar Mass Calculations - Introduction

Converting Between Moles, Atoms, and Molecules

Chemistry Reference table C
Step by Step Stoichiometry Practice Problems | How to Pass Chemistry
5.0 / 5 (0 votes)
Thanks for rating: