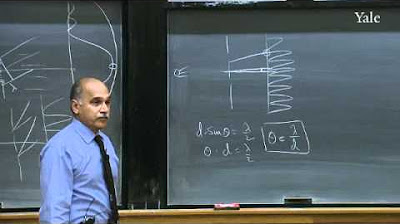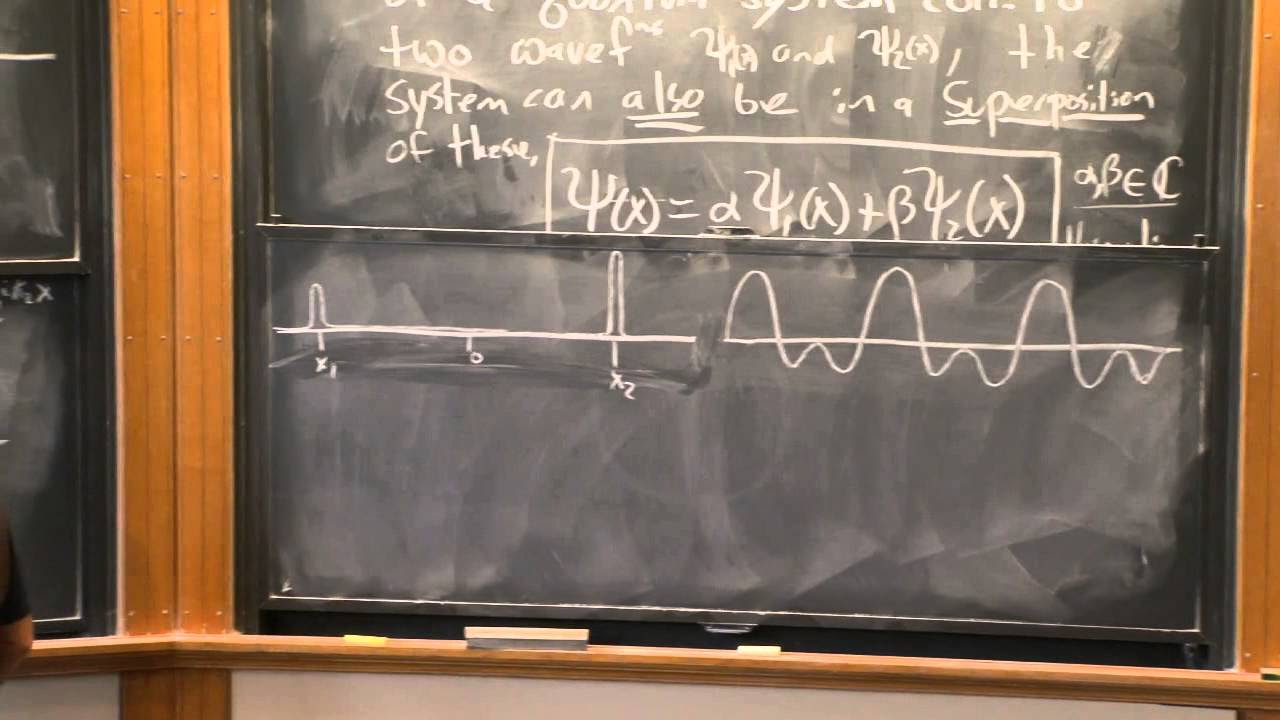The Heisenberg Uncertainty Principle Part 1: Position/Momentum and Schrödinger's Cat
TLDRThe video delves into the Heisenberg uncertainty principle, a fundamental concept in quantum mechanics that challenges classical determinism. It explains how the more precisely the position (delta x) of a particle is known, the less certain its momentum (delta p) becomes, and vice versa, with their product exceeding Planck's constant divided by 4π. This principle is not a limitation of our measuring tools but a core property of quantum particles, which exist as both particles and waves. The video also touches on the Copenhagen interpretation and the philosophical implications it brings, such as the idea of quantum superposition illustrated by Schrödinger's cat thought experiment, which questions the nature of reality and observation in a probabilistic universe.
Takeaways
- 📚 The Heisenberg uncertainty principle is a fundamental concept in quantum mechanics, indicating that precise values for both position and momentum of a quantum particle cannot be known simultaneously.
- 🔍 Quantum mechanics differs from classical mechanics in that it introduces probabilistic nature, rather than determinism, into the description of physical systems.
- 🌊 Particles in quantum mechanics exhibit wave-particle duality, which is central to understanding the probabilistic nature of their position and momentum.
- 🎲 The Copenhagen interpretation posits that an electron does not possess precise values for position and momentum until a measurement is taken, which results in a random outcome from a probability distribution.
- 📈 Heisenberg's uncertainty principle is mathematically expressed as the product of the uncertainties in position (Δx) and momentum (Δp) being greater than ħ/4π, where ħ is the reduced Planck constant.
- 🔄 The more precisely one parameter (position or momentum) is measured, the less precisely the other can be known, and vice versa.
- 🚫 The uncertainty principle is not a limitation of our measuring instruments but a fundamental property of quantum systems.
- 👁️ Observation in quantum mechanics has a tangible impact on the system being observed, as the act of measurement alters the state of the quantum system.
- 💡 Schrödinger's cat thought experiment was devised to challenge the Copenhagen interpretation and highlight the paradox of quantum superposition, where a system can exist in multiple states until observed.
- 🐱 The concept of quantum superposition suggests that, according to the Copenhagen interpretation, a quantum system exists in all possible states until it is observed, at which point it collapses into a single state.
- 🤔 Despite its initial intent to discredit the Copenhagen interpretation, Schrödinger's cat has become a classic thought experiment that continues to be discussed in the context of different interpretations of quantum mechanics.
Q & A
What is the Heisenberg uncertainty principle?
-The Heisenberg uncertainty principle states that when looking at complementary variables like position and momentum, the more precisely one parameter is known, the less we know about the other. It implies that it is impossible to simultaneously know the exact position and momentum of a quantum particle.
How does classical mechanics differ from quantum mechanics in terms of determinism?
-In classical mechanics, an object is believed to have precise values for its position and momentum at all times. However, in quantum mechanics, this determinism no longer applies because particles are also waves, and the realm is probabilistic in nature, not allowing for exact simultaneous knowledge of position and momentum.
What is the Copenhagen interpretation of quantum mechanics?
-The Copenhagen interpretation posits that an electron does not possess precise values for both position and momentum at the same time. Instead, when a measurement is taken, the result is randomly drawn from a probability distribution, and the act of measurement causes the system to collapse into one of the possible states.
How does the double-slit experiment demonstrate the wave-particle duality of electrons?
-The double-slit experiment shows that electrons can exhibit wave-like behavior, such as interference patterns, when not observed, but act as particles when their position is measured, thus demonstrating their dual nature as both waves and particles.
What is the relationship between the uncertainty in position (delta x) and momentum (delta p) as described by Heisenberg's principle?
-According to Heisenberg's uncertainty principle, the product of the uncertainties in position (delta x) and momentum (delta p) must be greater than h/(4π), where h is Planck's constant. If the uncertainty in one parameter decreases, the uncertainty in the other must increase.
What is the philosophical implication of a probabilistic universe as suggested by quantum mechanics?
-The idea that nature is not deterministic but probabilistic at its most fundamental level challenges our traditional understanding of cause and effect, and has profound implications for our understanding of reality, free will, and the nature of existence itself.
What is Schrodinger's cat thought experiment, and what does it illustrate?
-Schrodinger's cat is a thought experiment that illustrates the concept of quantum superposition. It involves a cat that is both alive and dead at the same time until an observation is made, at which point the cat collapses into a single state. The experiment was designed to highlight the absurdity of the Copenhagen interpretation but remains a classic example in the discussion of quantum mechanics interpretations.
How does the act of observation affect a quantum system?
-The act of observation in quantum mechanics has a concrete impact on the system. When a quantum system interacts with a photon or any other measuring device, the interaction itself alters the state of the system, demonstrating that observation is not a passive process but an active one that influences the outcome.
What was the reaction of the scientific community to the problems of measurement and determinism in quantum mechanics?
-The scientific community was initially in total confusion regarding the problems of measurement and the notion that nature is fundamentally probabilistic. Many scientists developed thought experiments to explore and sometimes challenge these concepts, leading to a deeper philosophical examination of quantum mechanics.
Why did Schrodinger create the cat paradox, and what was his intention?
-Schrodinger created the cat paradox to challenge and discredit the Copenhagen interpretation, as he found the idea of a superposition of states like being both dead and alive to be absurd. However, the paradox has since become a foundational element in discussions about the interpretations of quantum mechanics.
What is the significance of the Heisenberg uncertainty principle in the field of quantum mechanics?
-The Heisenberg uncertainty principle is a fundamental concept in quantum mechanics that highlights the probabilistic nature of the quantum realm. It has profound implications for our understanding of the limits of measurement and knowledge in the quantum world.
Outlines
🌟 Quantum Mechanics and Heisenberg's Uncertainty Principle
This paragraph introduces the Heisenberg uncertainty principle within the context of quantum mechanics. It explains the transition from classical mechanics, where objects have precise values for position and momentum, to quantum mechanics, where particles exhibit wave-particle duality and probabilistic behavior. The Copenhagen interpretation is introduced, highlighting that electrons do not possess precise values for both position and momentum simultaneously. The uncertainty principle is then defined, stating that the product of the uncertainties in position (delta x) and momentum (delta p) must be greater than h/4π, with one parameter's increased certainty leading to the other's increased uncertainty. The paragraph also touches on the philosophical implications of this principle and the concept of observation affecting the quantum system, as demonstrated by the double-slit experiment.
🐱 Schrödinger's Cat and Quantum Superposition
This paragraph discusses Schrödinger's cat thought experiment, which was devised to challenge the Copenhagen interpretation of quantum mechanics. It explains the concept of quantum superposition, where a quantum system can exist in multiple states until it is observed, at which point it collapses into a single definite state. The thought experiment involves a cat that is both alive and dead until the box it is in is opened and the cat's state is observed. The paragraph concludes by noting that while Schrödinger intended the paradox to discredit the Copenhagen interpretation, it remains a classic example in discussions of quantum mechanics and has led to various interpretations of the field.
Mindmap
Keywords
💡Heisenberg uncertainty principle
💡Quantum mechanics
💡Copenhagen interpretation
💡Wave-particle duality
💡Schrodinger equation
💡Probability distribution
💡Observation
💡Quantum superposition
💡Schrodinger's cat
💡Wave function
💡Planck's constant
Highlights
Professor Dave introduces the Heisenberg uncertainty principle.
Quantum mechanics developed by Schrodinger and others led to perplexing implications.
In classical mechanics, an object has precise values for position and momentum, but not in quantum mechanics.
Quantum particles are both particles and waves, leading to a probabilistic nature of the quantum realm.
The Copenhagen interpretation states that an electron does not possess precise values for position and momentum simultaneously.
Measurement results are randomly drawn from a probability distribution.
Heisenberg's uncertainty principle states that the more precisely one parameter is known, the less we know about the other.
The uncertainty in position (delta x) and momentum (delta p) must be greater than h/4π.
If the uncertainty in one parameter decreases, the uncertainty in the other must increase.
The limitations are not due to measuring instruments but are a fundamental quality of matter.
The double-slit experiment demonstrated that electrons are also waves.
Observation has a concrete impact on the quantum system.
The scientific community was confused by the problem of measurement in quantum mechanics.
The idea of a probabilistic universe had profound philosophical implications.
Schrodinger's cat thought experiment challenges the Copenhagen interpretation.
Quantum superposition suggests that a system can exist in multiple states until observed.
The cat in the box is in a superposition of dead and alive until the box is opened.
Schrodinger's paradox was intended to discredit the Copenhagen interpretation.
The thought experiment of Schrodinger's cat remains a classic in quantum mechanics discussions.
Transcripts
5.0 / 5 (0 votes)
Thanks for rating: