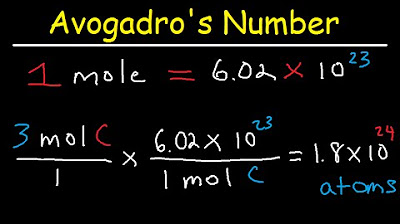How and Why Do Chemists Use Moles?
TLDRThis script explores the concept of 'moles' in chemistry, clarifying the confusion between the unit of measurement and the small mammals. It explains the use of moles as a convenient way to count atoms and molecules, introducing the Avogadro number, which represents the number of particles in one mole. The video also delves into historical experiments that laid the foundation for understanding molecular reactions and emphasizes the practical applications of moles in scaling chemical processes from the nanoscale to the macro scale, highlighting their importance in both theoretical and industrial chemistry.
Takeaways
- 🔬 A mole is a unit in chemistry used to count atoms and molecules, simplifying the process of dealing with very large numbers.
- 📏 The term 'mole' is not related to the animal but is a measure of quantity, similar to counting grains of rice in cups.
- 🌌 A mole represents 6.02214076 x 10^23 particles, a number known as Avogadro's number, which is agreed upon internationally.
- 📚 Avogadro's number, while named after the chemist Amedeo Avogadro, was not defined by him but rather by the International Bureau of Weights and Measures.
- 🧪 Moles help bridge the macroscopic and microscopic scales, allowing chemists to work with manageable numbers in both large and small scales.
- 🔄 The concept of moles allows for the conversion between the number of molecules and the mass of a substance through molar mass.
- 🌡️ At standard temperature and pressure, one mole of a gas occupies 22.4 liters, a principle used to understand gas reactions.
- 🔬 Avogadro was the first to suggest that equal volumes of gases, at the same temperature and pressure, contain the same number of molecules, regardless of the type of gas.
- 🛠️ Moles are used in experiments to determine the quantities of reactants and products in chemical reactions, such as the reaction between hydrogen and oxygen to form water.
- 🧪 Chemists use moles to weigh out the correct amounts of solid substances for experiments, based on their molar masses.
- 🌐 Moles and Avogadro's number are essential for understanding and designing systems that operate at the nanoscale, such as in the development of new energy technologies.
Q & A
What is the purpose of using moles in chemistry?
-Moles are used in chemistry to simplify the counting of atoms, molecules, or other entities in a chemical reaction. Instead of dealing with extremely large numbers, chemists can use moles, which are based on Avogadro's number, to make calculations more manageable.
What is the Avogadro's number and why is it significant in chemistry?
-Avogadro's number is approximately 6.02214076 x 10^23 and represents the number of particles (atoms, molecules, etc.) in one mole of a substance. It is significant because it allows chemists to relate the macroscopic quantities of substances to the number of particles at the atomic or molecular level.
Why is the mole concept useful when dealing with gases?
-The mole concept is particularly useful for gases because it allows chemists to relate the volume of a gas to the number of particles it contains. At standard temperature and pressure, one mole of any gas occupies the same volume, which is 22.4 liters.
How can the concept of moles help in understanding chemical reactions?
-Moles help in understanding chemical reactions by providing a way to quantify the reactants and products in terms of their particle numbers. This makes it possible to balance chemical equations and predict the amounts of substances produced or consumed in a reaction.
What is the relationship between moles and mass in chemistry?
-The relationship between moles and mass is given by the molar mass of a substance, which is the mass of one mole of that substance. By knowing the molar mass, one can calculate the mass of a given number of moles of a substance or vice versa.
How does the mole concept help in scaling up chemical reactions from the laboratory to industrial scale?
-The mole concept allows chemists to scale up chemical reactions by providing a consistent way to measure and compare quantities of substances regardless of the scale. This is essential for industrial processes where large volumes of products are required.
What is the historical significance of Avogadro's hypothesis in the development of chemistry?
-Avogadro's hypothesis, which states that equal volumes of gases at the same temperature and pressure contain the same number of molecules, was pivotal in establishing the molecular theory of matter. It laid the groundwork for understanding the discrete nature of chemical reactions and the concept of moles.
How does the mole concept relate to the measurement of electrical quantities such as voltage and current?
-The mole concept can be used in conjunction with Avogadro's number to relate macroscopic electrical measurements to the behavior of individual electrons or other charge carriers at the nanoscale, thus bridging the gap between macroscopic and microscopic phenomena.
What is the role of moles in preparing chemical solutions and buffers?
-Moles are used to determine the correct amounts of solute to dissolve in a given volume of solvent to achieve a specific molar concentration. This is crucial for preparing chemical solutions and buffers with precise concentrations for experimental purposes.
How can the mole concept be used to understand and design new power systems like hydrogen fuel cells?
-By using the mole concept, chemists can calculate the amounts of reactants and products involved in electrochemical reactions, such as those in hydrogen fuel cells. This helps in understanding the reaction kinetics and designing more efficient energy systems.
Outlines
🔬 Understanding Moles in Chemistry
This paragraph introduces the concept of moles in chemistry, which are a way to count atoms and molecules using the Avogadro number, approximately 6.02214076 x 10^23. It compares counting moles to measuring ingredients in cooking, emphasizing the impracticality of counting individual atoms. The speaker humorously clarifies the difference between 'molds' and 'moles' and explains the necessity of using moles to simplify calculations involving vast numbers of atoms and molecules.
📏 Scaling from Nano to Macro: Avogadro's Number
The speaker explains the importance of Avogadro's number in bridging the gap between the nanoscale of individual atoms and molecules and the macroscale of measurable quantities in chemistry. It discusses the role of the International Bureau of Weights and Measures in defining the Avogadro number and how it is used to convert between the number of molecules and the volume of gases at standard temperature and pressure.
🔍 Historical Foundations of Chemistry and Avogadro's Contributions
This paragraph delves into the historical context of Avogadro's hypothesis, which states that equal volumes of gases at the same temperature and pressure contain the same number of molecules. The speaker describes how this principle was foundational to early chemical experiments and the development of the field, leading to the understanding of atoms and molecules as the building blocks of matter.
🌡️ Gas Laws and the Molecular Perspective
The speaker uses the example of a hydrogen and oxygen gas reaction to illustrate how early chemists used macro-scale experiments to deduce molecular behavior. It explains the consistent volume ratios in chemical reactions, such as the formation of water from hydrogen and oxygen, which led to the understanding of molecular proportions and the validation of Avogadro's hypothesis.
🧪 Practical Applications of Moles in Chemistry
This paragraph discusses the practical use of moles in preparing chemical experiments, particularly with solids. It provides an example of how to calculate the necessary amounts of chemicals for an experiment using molar masses and the Avogadro number. The speaker also touches on the broader applications of moles in various fields, including the study of catalysts and the development of industrial processes.
🔋 Moles and the Connection Between Macro and Nano Scales
The final paragraph emphasizes the utility of moles in translating between macro-scale measurements and nanoscale phenomena. It highlights how moles allow chemists to design and scale up processes from the laboratory to industrial applications, such as hydrogen fuel cells. The speaker also hints at a forthcoming video that will explore the origins of the Avogadro number in more detail.
📢 Closing Remarks and Invitation for Engagement
In the closing paragraph, the speaker invites viewers to engage with the content by asking questions or leaving comments and encourages them to subscribe for future videos. There is a playful note as the speaker jokingly calls viewers back to work after the video ends, adding a light-hearted touch to the conclusion.
Mindmap
Keywords
💡Mole (in chemistry)
💡Avogadro's number
💡Macro scale
💡Nano scale
💡Molar mass
💡Gas laws
💡Chemical reactions
💡Stoichiometry
💡Buffers
💡Catalysts
💡Mole concept
Highlights
The concept of 'moles' in chemistry is used to simplify the counting of atoms and molecules, which would otherwise involve unimaginably large numbers.
A mole is a unit that represents a specific, incredibly large number of particles, approximately 6.02214076 x 10^23, known as Avogadro's number.
The use of moles allows chemists to work with more manageable numbers when dealing with atoms and molecules.
Avogadro's number was established by the International Bureau of Weights and Measures to standardize scientific measurements.
Moles provide a bridge between the macro scale, where measurements are made, and the nano scale, the realm of individual atoms and molecules.
Amadeo Avogadro, a 19th-century chemist, hypothesized that equal volumes of gases at the same temperature and pressure contain the same number of molecules.
Chemical experiments with gases, such as the reaction between hydrogen and oxygen to form water, helped establish the existence of molecules.
Understanding the relationship between gas volumes in reactions was key to the development of atomic theory.
Moles enable the calculation of the number of molecules in a given volume of gas at standard temperature and pressure.
The molar mass of a substance can be determined by adding the atomic masses of its constituent atoms and multiplying by Avogadro's number.
Moles are used not only for atoms and molecules but also for other entities like photons or electrons that relate to the nanoscale.
Moles facilitate the translation between macro-scale measurements like voltage and current to nanoscale phenomena involving atoms.
Chemists use moles to weigh out the correct amounts of solid substances for experiments, based on their molar masses.
The concept of moles is essential for scaling chemical reactions from the laboratory to industrial applications.
Moles are used to understand and design systems like hydrogen fuel cells, which operate on the principles of molecular interactions.
The use of moles simplifies calculations involving vast numbers of particles, making chemistry more accessible and practical.
A video series will explore the historical development of Avogadro's number and its significance in chemistry.
Transcripts
Browse More Related Video
5.0 / 5 (0 votes)
Thanks for rating: