A Simple Distillation Explained
TLDRThis instructional video guides viewers through building a simple still apparatus for distillation. It covers the setup process, including assembling the round bottom flask, three-way connector, condenser, and thermometer, as well as the use of a lab jack, stir plate, and thermal well for heating. The script explains the importance of gas-tight seals, temperature monitoring, and even heating to avoid bumping. It concludes with the distillation process, emphasizing the gradual change in distillate composition and the collection of data points to determine when to stop.
Takeaways
- 🔬 The video is a tutorial on building a simple still apparatus for distillation.
- 🔧 Essential equipment includes a round bottom flask, a three-way connector, a thermometer adapter, a condenser, and a vacuum adapter.
- 🔨 A lab jack, stir plate, and thermal well are used for setup and stirring, with the stir plate serving as a hot plate in this case.
- ⚙️ The thermometer should be snugly fitted with vacuum grease to ensure a gas-tight seal.
- 🔄 A vacuum grease is used for creating gas-tight seals at the joints of the apparatus.
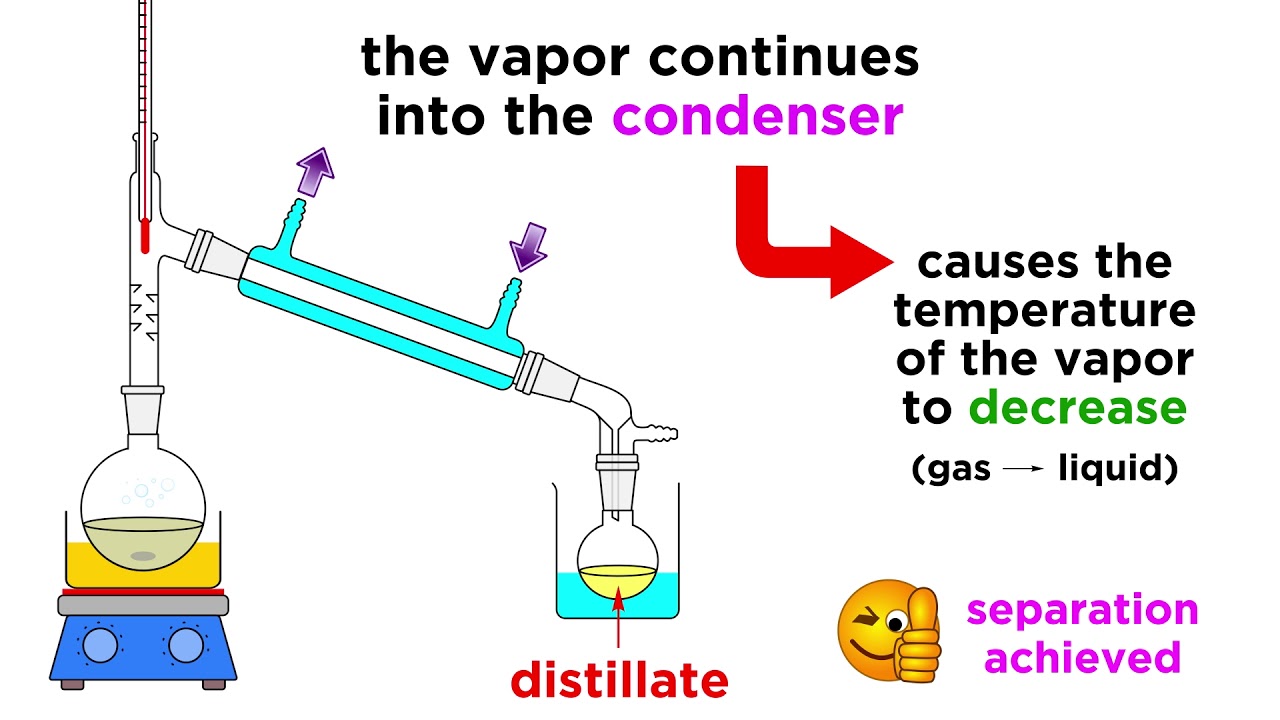
- 💧 A West condenser with a vacuum adapter is used, and cooling water is applied to it for efficient condensation.
- 📏 A graduated cylinder is used to collect and measure the volume of the distillate.
- 🔄 The setup involves clamping and adjusting the position of various components to ensure proper alignment and sealing.
- 🌡 Monitoring the temperature in the still head is crucial for controlling the distillation process.
- 🌡️ The temperature in the still head increases as distillation progresses due to the changing composition of the distillate.
- 📈 Plotting temperature against the volume of distillate collected helps in understanding the distillation process and determining when to stop.
- 🛑 The distillation process should be stopped when about half of the original volume has been distilled, indicating that most of the ethanol has been removed.
Q & A
What is the purpose of the round bottom flask in the script?
-The round bottom flask is used as the primary container for the mixture being distilled. It is heated to vaporize the components of the mixture.
What is the role of the three-way connector in the setup described?
-The three-way connector is used to connect different parts of the distillation apparatus, such as the still head and the flow director, ensuring a gas-tight seal for efficient distillation.
Why is vacuum grease used in the script?
-Vacuum grease is applied to the joints of the glassware to ensure a gas-tight seal, preventing the escape of vapors and maintaining the integrity of the distillation process.
What is the function of the West condenser in the distillation setup?
-The West condenser is used to cool the vapor coming from the still head, causing it to condense back into a liquid form, which then collects in the receiver.
Why is a thermometer adapter used in the script?
-The thermometer adapter is used to securely hold the thermometer in place, allowing for accurate temperature monitoring of the vapor during the distillation process.
What is the purpose of the vacuum adapter mentioned in the script?
-The vacuum adapter is used to create a vacuum or reduced pressure environment, which can facilitate the distillation process by lowering the boiling point of the mixture.
What is the role of the stir plate in the distillation process described?
-The stir plate is used to stir the mixture in the round bottom flask, ensuring even heating and preventing the formation of 'bumps' or uneven boiling.
How does the script ensure an even boil in the distillation process?
-The script describes adjusting the stir plate speed and thermal well temperature to achieve an even boil, which is crucial for the efficient separation of components during distillation.
What is the significance of monitoring the temperature in the still head as a function of the distillate collected?
-Monitoring the temperature helps to determine the boiling point of the distillate and indicates the composition changes as the distillation progresses, which is important for the separation of components.
Why is it important to know the exact volume of distillate collected as mentioned in the script?
-Knowing the exact volume of distillate is important for accurate record-keeping and for understanding the efficiency of the distillation process, as well as for any subsequent analysis or use of the distillate.
How does the script describe the process of stopping the distillation?
-The script suggests stopping the distillation when about half of the total volume added to the flask has been collected and when the temperature in the still head levels off, indicating that most of the ethanol has been removed.
Outlines
🔬 Setting up a Basic Distillation Apparatus
This paragraph outlines the initial setup for a simple distillation process. The narrator discusses the necessary equipment, including a round bottom flask, a three-way connector, a steel head, a thermometer adapter, a West condenser, a vacuum adapter, and a graduated cylinder. They also mention the need for a lab jack, a stir plate, a thermal well as a heat source, and various clamps and connectors. The focus is on assembling the apparatus correctly, ensuring gas-tight seals with vacuum grease, and positioning the equipment for optimal distillation.
🌡️ Conducting the Distillation and Collecting Data
In this paragraph, the narrator proceeds with the distillation process. They describe the use of a beaker for elevation and a graduated cylinder for collecting the distillate, emphasizing the importance of accurately measuring the volume. The setup includes applying cooling water to the condenser, loading the sample mixture of ethanol and water into the round bottom flask, and using a spin bar for even heating. The process involves monitoring the temperature in the still head and adjusting the stir plate and thermal well settings for steady heating and stirring. The narrator explains how to observe the condensation and collection of vapor, and how to plot temperature against distillate volume to determine when to stop the distillation once the ethanol has been mostly removed.
Mindmap
Keywords
💡Still
💡Three-way connector
💡Thermometer adapter
💡Vacuum adapter
💡Graduated cylinder
💡Lab jack
💡Stir plate
💡Thermal well
💡Vacuum grease
💡Condenser
💡Distillate
Highlights
Introduction to building a simple still apparatus for distillation.
List of required equipment including round bottom flask, three-way connector, and thermometer adapter.
Use of a West condenser and vacuum adapter for directing flow into a graduated cylinder.
Necessity of connectors, clamps, and a lab jack for setting up the apparatus.
Utilization of a stir plate as a hot plate for stirring the mixture.
Connecting the stir plate to a non-variable power outlet and the thermo L to a variable power outlet.
Securing the round bottom flask with a clamp and adjusting the lab jack for proper positioning.
Applying vacuum grease for a gas-tight seal when connecting the three-way adapter.
Placing the flow director and condenser with attention to positioning and gas-tight connections.
Using a thermometer adapter with vacuum grease to monitor the temperature in the field.
Securing the condenser with a ket clip for a snug connection without clamps.
Selection of a beaker and graduated cylinder for collecting distillate and monitoring volume.
Connecting a flexible water hose for cooling the condenser with proper water flow direction.
Loading the sample mixture into the boiling flask and adding a spin bar for even heating.
Activating the fume hood, stir plate, and thermal well for controlled distillation.
Observation of vapor condensing and the importance of adjusting stirring and heating rates.
Monitoring temperature changes and distillate composition throughout the distillation process.
Identifying the endpoint of distillation when ethanol has been mostly removed.
Stopping the distillation process once half of the original volume has been distilled.
Transcripts
Browse More Related Video
5.0 / 5 (0 votes)
Thanks for rating: