What Are Chemical Properties? | Chemistry Matters
TLDRIn 'Chemistry Matters,' the exploration shifts from physical to chemical properties of matter, highlighting how elements react to form compounds vital for life. The video delves into chemical changes, illustrating through examples like combustion and oxidation, and distinguishes between physical and chemical properties. It uses demonstrations with magnesium, flash paper, and methyl alcohol to clarify concepts, emphasizing the importance of understanding these changes in the study of chemistry.
Takeaways
- 🔬 Chemical properties are characteristics of substances that are observed during chemical reactions, unlike physical properties which do not involve a change in the substance's chemical makeup.
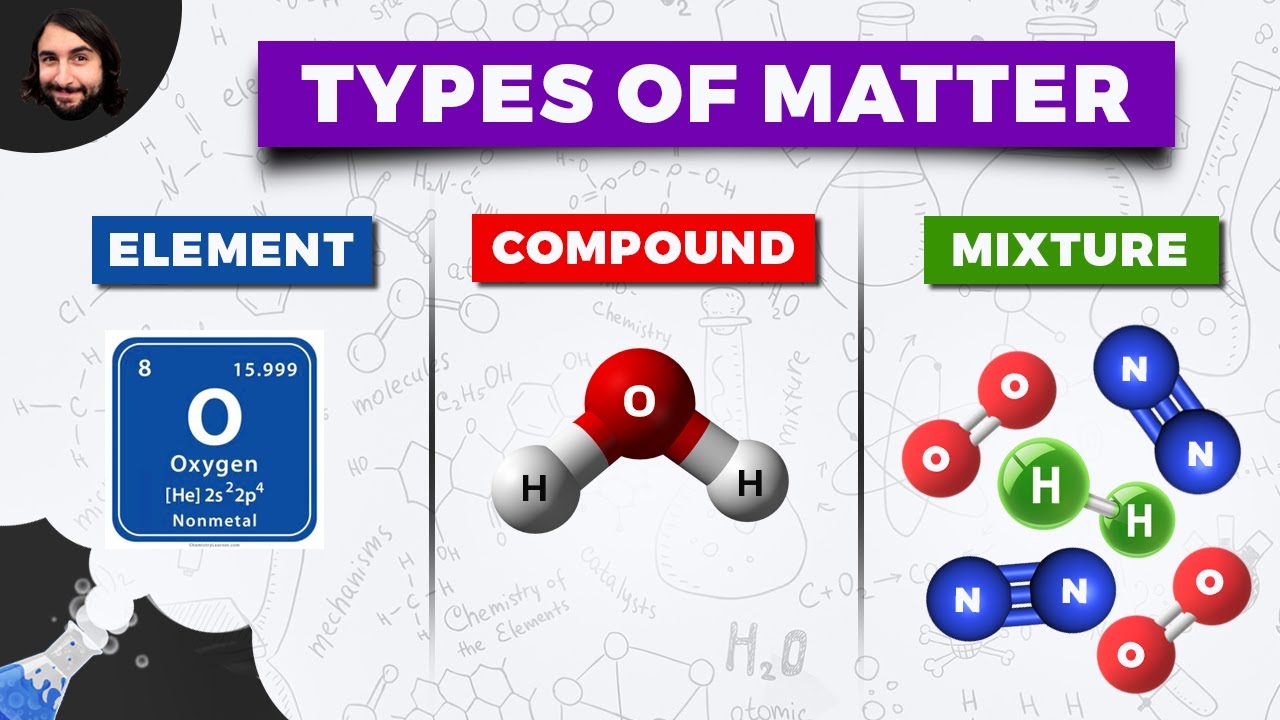
- 🧪 Most matter in nature exists as mixtures or combinations of elements, and chemical changes result in the formation of new chemical substances.
- 🔄 A compound is a substance formed from two or more elements chemically joined together, and while they do not change through physical changes, they can be separated by chemical changes.
- 🔥 Flammability and combustibility are chemical properties; a combustible material catches fire at a temperature above 43 degrees Celsius, while a flammable material ignites at a lower temperature.
- 🔥 Wood and candles are examples of combustible materials, whereas isopropyl alcohol is an example of a flammable material, both undergoing combustion when reacting with oxygen.
- ⚗️ Reactivity is a key chemical property, describing the tendency of substances to undergo chemical reactions, with metals like magnesium reacting quickly with oxygen to form compounds.
- 🎨 Corrosion or oxidation is an example of slow reactivity in metals, as seen with the Statue of Liberty's change in color due to oxidation of copper.
- 🥞 Cooking pancakes is a chemical change, as it involves a transformation that cannot be reversed, indicated by color change, release of heat, and formation of new substances.
- 🌫️ Combustion, as seen with flash paper, is a chemical change characterized by the release of energy, often in the form of light and heat.
- 🧊 The phase change from liquid to gas, such as vaporization, and from gas to liquid, such as condensation, are physical changes that involve a change in state but not in chemical composition.
- 🍬 A mixture is a combination of two or more pure substances where each retains its individual chemical properties, akin to the individual characteristics of elements in the atmosphere.
Q & A
What is a chemical property?
-A chemical property is a characteristic of a substance that is observed during a chemical reaction, such as flammability or reactivity.
How is a compound formed?
-A compound is formed when two or more elements combine chemically to create a new substance with its own unique properties.
What is the difference between physical and chemical changes?
-Physical changes involve a change in the state or appearance of a substance without altering its chemical composition, while chemical changes result in the formation of new substances with different chemical properties.
What is flammability?
-Flammability is the ability of a material to catch fire at a temperature above 43 degrees Celsius or 100 degrees Fahrenheit.
What is the example of a combustible material mentioned in the script?
-Wood is given as an example of a combustible material in the script.
What is the difference between a combustible and a flammable material?
-A combustible material catches fire at a higher temperature, while a flammable material ignites at a lower temperature and burns vigorously.
What is the chemical reaction between a candle and oxygen called?
-The chemical reaction between a candle and oxygen is called combustion.
What is reactivity in the context of chemical properties?
-Reactivity refers to the relative ability of a substance to undergo a chemical reaction, either by combining with other substances or breaking apart.
What is an example of slow reactivity in metals?
-Corrosion or oxidation, such as the transformation of the Statue of Liberty's copper to a green color, is an example of slow reactivity in metals.
How does aluminum protect itself from further oxidation?
-Aluminum reacts with oxygen in the air to form a protective layer of aluminum oxide, which prevents further oxidation.
What are the signs that a chemical change has occurred?
-Signs of a chemical change include a color change, the release of light or heat, the formation of a gas, and sometimes the formation of a new solid substance called a precipitate.
What is a mixture in chemistry?
-A mixture is a combination of two or more pure substances where each substance retains its individual chemical properties.
How does the script differentiate between vaporization and condensation?
-Vaporization is the phase change from a liquid to a gas, while condensation is the change from a gas back to a liquid.
Why does the script mention that methyl alcohol boils at human body temperature?
-The script mentions this to illustrate that methyl alcohol has weak intermolecular forces, allowing it to vaporize easily at a relatively low temperature.
Outlines
🧪 Introduction to Chemical Properties and Reactions
This paragraph introduces the concept of chemical properties, which are characteristics of substances that become evident during chemical reactions. It explains that elements in nature rarely exist alone and often combine to form compounds through chemical changes. A compound is a substance composed of two or more elements chemically bonded together. The paragraph also distinguishes between physical and chemical properties, with the latter only observable during chemical reactions. It uses examples such as flammability to illustrate chemical properties and discusses the importance of reactivity, showing how metals like magnesium react with oxygen to form compounds. The segment ends with a demonstration of magnesium burning, highlighting the energy release during chemical reactions.
🔍 Distinguishing Between Physical and Chemical Changes
This paragraph delves into the differences between physical and chemical changes through a series of examples and demonstrations. It starts with a discussion on the combustion of flash paper, which is identified as a chemical change due to the release of energy and the formation of smoke. The conversation then moves to cooking pancakes, which is also a chemical change, evidenced by the irreversible transformation from batter to a cooked state, color changes, and the release of heat and gases. Another example involves a liquid that undergoes vaporization and condensation, demonstrating a physical change as the substance returns to its original state. The paragraph concludes with an exploration of mixtures, comparing them to elements in the atmosphere that retain their individual properties, and teases further learning about mixtures in upcoming videos.
Mindmap
Keywords
💡Chemical Property
💡Chemical Change
💡Compound
💡Combustion
💡Reactivity
💡Oxidation
💡Corrosion
💡Physical Change
💡Phase Change
💡Mixture
💡Intermolecular Force
Highlights
Introduction to the concept of chemical properties, which are observed during chemical reactions.
Explanation that elements rarely exist alone in nature and often undergo chemical changes.
Definition of a chemical change as any change resulting in the formation of a new chemical substance.
Description of compounds as substances formed from two or more chemically joined elements.
Clarification that compounds can only be separated by chemical changes, not physical ones.
Illustration of the necessity of a chemical reaction to observe a chemical change.
Discussion on the definition of physical property and its contrast with chemical properties.
Flammability as an example of a chemical property, with the distinction between combustible and flammable materials.
Demonstration of wood and candle as examples of combustible materials and their chemical reactions.
Introduction of reactivity as a key chemical property, with metals being highly reactive.
Visual demonstration of magnesium reacting with oxygen to form magnesium oxide.
Explanation of slow reactivity through the example of the Statue of Liberty's oxidation.
Discussion on the prevention of corrosion using protective coatings on metals.
Identification of signs of chemical changes, such as color change, light or heat release, and gas formation.
Differentiation between physical and chemical changes using examples like flash paper and cooking pancakes.
Introduction to phase changes, such as vaporization and condensation, and their distinction from chemical changes.
Application of concepts learned about chemical changes to identify them in various situations.
Introduction to the concept of mixtures and their composition of pure substances retaining individual properties.
Transcripts
Browse More Related Video

1.1 Introduction to High School Chemistry and Matter | High School Chemistry

Changes in the Properties of Matter Physical and Chemical

Physical and Chemical Changes for Kids
Types of Matter: Elements, Compounds, and Mixtures

Atoms and Matter for Kids

Introduction for Reactions & Equations - AP Chem Unit 4, Topics 1-2a
5.0 / 5 (0 votes)
Thanks for rating: