The Electron: Crash Course Chemistry #5
TLDRThis video explains the concept of electron orbitals and their importance in chemistry. It discusses the history of models like Bohr's that tried to explain electron behavior. It uses a musical metaphor to explain how electrons exist in discrete energy levels or 'harmonics.' The periodic table orders elements by electron configuration. The script explains orbitals (s, p, d, f) as the 'notes' electrons can occupy. Filling orbitals leads to stability. The script argues that grasp of orbitals, not planets circling a nucleus, is key to visualizing atoms. Orbitals shape reactivity; their excitation makes existence possible.
Takeaways
- 📖 Mendeleev's periodic table discovery was influenced by the then-unknown properties of electrons, debunking his mystical views on element organization.
- 🎶 John Newlands' musical analogy for element periodicity, though ridiculed at the time, hinted at the deeper truths of element relationships and quantum mechanics.
- 🔬 Niels Bohr's model of the atom introduced the concept of quantized energy levels, laying groundwork for understanding electron behavior despite its limitations.
- 🤖 Electrons are better described as waves rather than particles, challenging traditional visualizations of atoms with orbiting electrons.
- 🎵 Quantum mechanics views electrons in terms of wave-particle duality, introducing the concept of electron harmonics and standing waves around the nucleus.
- 📈 Electron orbitals and shells organize electrons in atoms, influencing chemical properties and reactions based on the number and arrangement of electrons.
- 🚨 The octet rule, describing atoms' preference for eight electrons in their outer shell, resembles musical harmony, emphasizing stability and reactivity.
- 🖥 Electron configurations provide a way to describe the distribution of electrons in atoms, crucial for understanding chemical behavior.
- 💡 Ionization energy and electron affinity reflect the energy changes associated with adding or removing electrons, influencing chemical reactivity.
- 📚 The periodic table's arrangement encapsulates electron configurations, elemental stability, and the fundamental principles governing chemical interactions.
Q & A
Who was John Newlands and what was his contribution to the periodic table?
-John Newlands was a young chemist and activist who in 1865 published a paper noting the periodicity of elements by comparing them to a musical scale. He theorized that elements might repeat in 'octaves' like notes in music.
Why was Niels Bohr important in developing theories about atomic structure?
-Niels Bohr was a Danish physicist who in 1913 came up with a simple model for describing electron energy levels in hydrogen atoms. This laid important groundwork for quantum mechanics and modeling atomic structure.
What is the difference between orbiting electrons as particles versus electrons as waves?
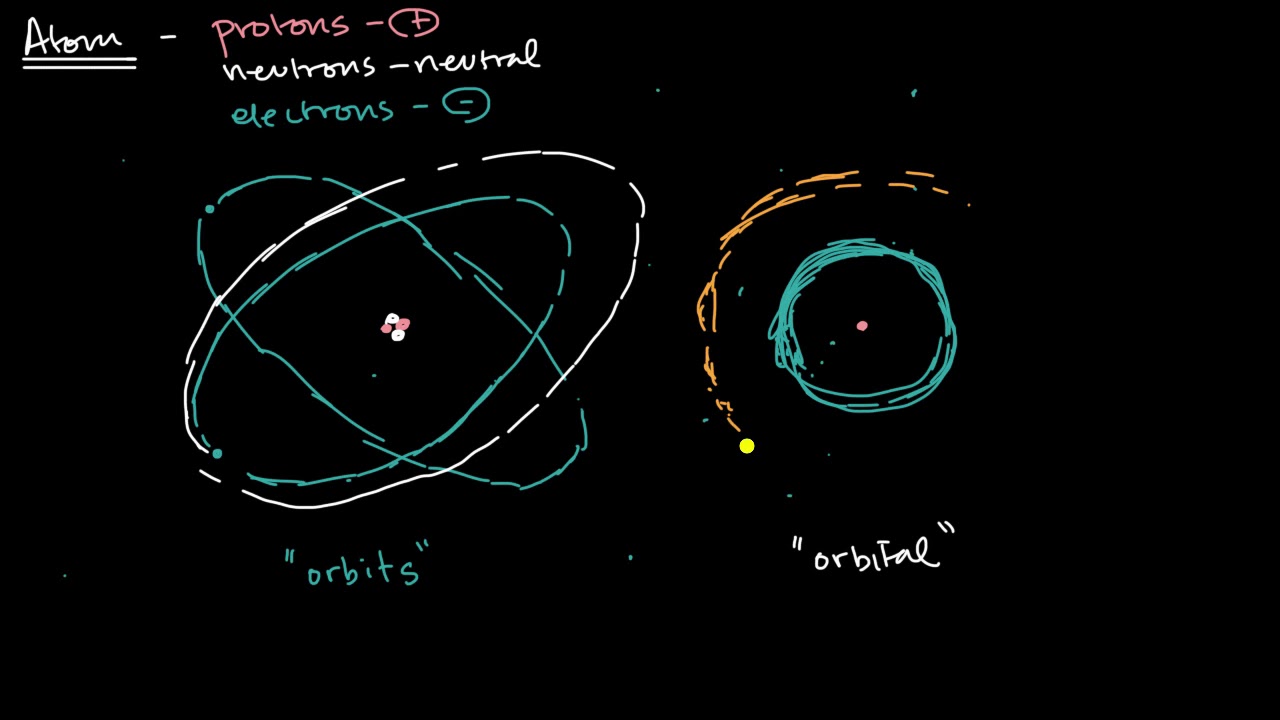
-The original model saw electrons as solid particles orbiting the nucleus, but it was discovered that electrons behave more like waves. Electron waves exist in 'orbitals' around the nucleus in certain energy levels and configurations.
What are the different types of orbitals that exist in electron shells?
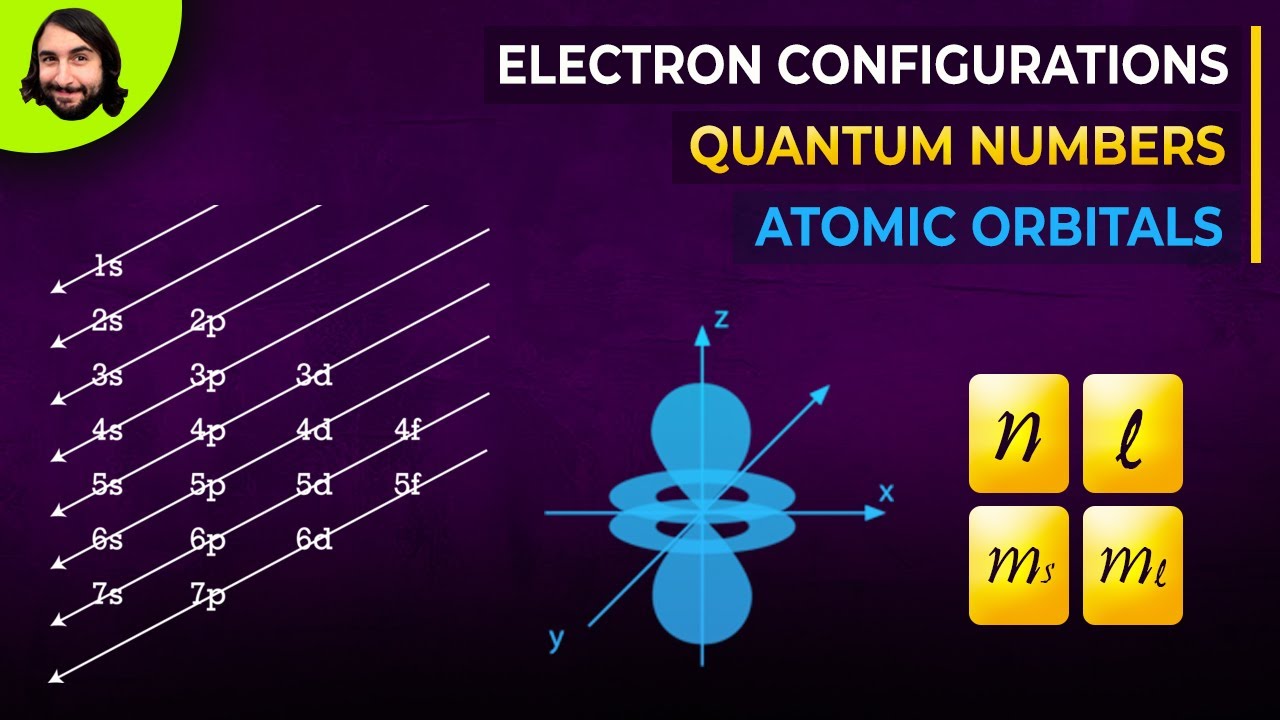
-The main orbital types are: s-orbitals which can hold 2 electrons, p-orbitals which are 3-part harmonies holding 6 electrons, and d-orbitals which are 5-part harmonies holding 10 electrons.
How are electron configurations written out?
-Electron configurations list the shell number, orbital letter, and electrons in that orbital in sequence. For example, fluorine is written as 1s2 2s2 2p5.
What causes the diagonal relationship in filling electron orbitals?
-Filling the 3d orbital requires a lot of energy, so electrons go into the 4s orbital first before filling the 3d, causing the diagonal pattern.
What are ionization energy and electron affinity?
-Ionization energy is the discrete energy required to remove an electron from an atom. Electron affinity is the energy associated with adding an electron to fill an orbital.
How do the orbitals relate to reactivity and conductivity?
-Incomplete outer orbitals make atoms more reactive as they seek to fill them. Inner d/f orbitals are shielded and less reactive, allowing those elements to conduct electricity.
How does the periodic table reflect electron configurations?
-The table groups elements by orbital filling patterns. The rows match the maximal electrons in s, p, d, f orbitals. This shows reactivity patterns.
What is the electron field and how do electron waves relate to probability?
-The electron field permeates the universe. Electron waves are excitations in this field, with the wave strength at a point determining the probability of finding an electron there.
Outlines
😃 Introducing Hank Green and Crash Course Chemistry
Hank Green welcomes viewers to Crash Course Chemistry. He talks about how Mendeleev discovered periodicity in elements but didn't understand electrons. Hank says that by the end of the video, viewers will understand electrons.
😮 Earlier models of atomic structure
Hank discusses earlier models of atomic structure by Newlands and Bohr. Newlands compared periodicity of elements to music but was laughed off. Bohr created a too-simple model of electrons orbiting nucleus like planets. His model failed for complex atoms because electrons behave more like waves.
🎵 Understanding electrons through music
Hank explains how he thinks of electrons in musical terms - as vibrations and harmonies. He talks about standing electron waves, orbitals as musical notes, incomplete orbitals causing disharmony, and noble gases achieving harmony. He then explains how to write electron configurations.
Mindmap
Keywords
💡Electrons
💡Orbitals
💡Electron shells
💡Periodic table
💡Noble gases
💡Ionization energy
💡Electron affinity
💡Quantum mechanics
💡Electron configurations
💡Octet rule
Highlights
The research uses an innovative combination of computational modeling and neuroimaging to understand decision making.
By comparing model predictions to actual brain activity, the study provides new insights into how different brain regions interact during choice behaviors.
The key theoretical contribution is a novel framework integrating both algorithmic models from psychology and implementation models from neuroscience.
The model makes accurate quantitative predictions of behavior and brain activity in multiple brain regions during simple value-based choices.
The results demonstrate the orbitofrontal cortex encodes economic subjective values for different options during decision making.
The model suggests how psychological constraints and biological implementations interact to shape choice behavior at the algorithmic level.
The researchers identify a candidate neural mechanism for how the brain computes and compares subjective values to make choices.
The framework integrates the interactions between valuation, attention, and choice processes at both algorithmic and implementation levels.
The model provides a new computational account of the neural basis of simple economic choice in the human brain.
The study has important practical implications for understanding and treating disorders of decision making like addiction.
The modeling approach could be extended to study more complex decisions and inform treatments for impaired decision making.
The combined computational modeling and neuroimaging approach provides a powerful framework to dissect the mechanisms underlying choice.
The model makes falsifiable predictions linking neural computations to behavior that can guide and constrain future research.
This innovative fusion of theory, modeling, and data moves us closer to a computational understanding of decision making.
The study provides an important step toward translating our understanding of decision neuroscience into biomedical applications.
Transcripts
Browse More Related Video

Orbitals, the Basics: Atomic Orbital Tutorial — probability, shapes, energy |Crash Chemistry Academy
Shells, subshells, and orbitals | Atomic structure and properties | AP Chemistry | Khan Academy

How to Write the Electron Configuration of an Element | Study Chemistry With Us

More on orbitals and electron configuration | Chemistry | Khan Academy
Quantum Numbers, Atomic Orbitals, and Electron Configurations

BTEC Applied Science: Unit 1 Chemistry Electron Configuration
5.0 / 5 (0 votes)
Thanks for rating: