The Mole: Avogadro's Number and Stoichiometry
TLDRProfessor Dave's video script delves into the concept of moles in chemistry, a fundamental unit that allows chemists to deal with the vast number of molecules that are too small to see individually. A mole is defined as Avogadro's number, approximately 6.022 x 10^23, representing the number of carbon atoms in 12 grams of carbon. It bridges the gap between atomic mass units and grams, enabling chemists to perform stoichiometric calculations. The script explains how to convert between mass and moles using molar mass, and demonstrates this with a practical example of a combustion reaction involving propane and water. The process emphasizes the importance of unit cancellation and the predictive power of chemistry in technological advancements. The summary encourages viewers to subscribe for more educational content and to reach out with any questions.
Takeaways
- 🔬 A mole is a way to numerically represent a large number of molecules that we can observe and work with in chemistry.
- 📏 The number of molecules in a mole is Avogadro's number, which is approximately 6.022 x 10^23.
- 🌟 A mole is defined as the number of carbon atoms in exactly 12 grams of carbon-12, which is a tangible amount for chemical experiments.
- ⚖️ The molar mass of a substance is the same as its molecular mass but expressed in grams instead of atomic mass units.
- 💧 For example, a mole of water molecules weighs 18 grams, which is the sum of the atomic masses of its constituent atoms.
- 🔢 Stoichiometry allows us to predict the quantities involved in chemical reactions by using mole ratios.
- 🔥 In the example given, starting with 20 grams of propane, we can calculate the mass of water produced through stoichiometric calculations.
- 🧮 To convert between mass and moles, we use the molar mass of the substance, which is a key step in stoichiometric calculations.
- 📉 Multiplying the number of moles of a substance by its stoichiometric coefficient gives the expected number of moles of the product.
- ↔️ When performing calculations, it is crucial to ensure that units cancel out appropriately to confirm the correctness of the calculation.
- 📚 Stoichiometric calculations are powerful tools in chemistry that enable us to make precise predictions and contribute to technological advancements.
- 💌 The script encourages viewers to subscribe for more tutorials and to reach out with questions via email.
Q & A
What is the significance of Avogadro's number in chemistry?
-Avogadro's number, which is approximately 6.022 x 10^23, is significant because it represents the number of particles, such as molecules or atoms, in one mole. This allows chemists to relate the microscopic scale of particles to a macroscopic scale that can be measured and observed.
How does the mole concept help in stoichiometry?
-The mole concept helps in stoichiometry by providing a way to express the number of molecules or atoms in a sample numerically. It allows chemists to use whole numbers in balancing chemical equations and performing calculations, which would otherwise involve very small or large numbers that are impractical to work with.
What is the relationship between atomic mass units and grams in the context of molar mass?
-The molar mass of an element or compound is the mass of one mole of that substance, expressed in grams. It is numerically equal to the atomic or molecular mass in atomic mass units (amu), but the units are grams per mole. This relationship allows chemists to convert between mass and the number of moles of a substance.
How is the molar mass of a compound calculated?
-The molar mass of a compound is calculated by adding the atomic masses of all the atoms present in one molecule of the compound, as listed on the periodic table. These atomic masses are then summed to give the total molar mass of the compound.
What is the molar mass of water in grams per mole?
-The molar mass of water (H2O) is 18 grams per mole, which is calculated by adding the atomic mass of two hydrogen atoms (1 amu each) and one oxygen atom (16 amu).
How can you convert the mass of a substance to the number of moles?
-To convert the mass of a substance to the number of moles, you divide the mass of the substance by its molar mass. The molar mass is the mass of one mole of the substance and is usually expressed in grams per mole.
What is the stoichiometric ratio in a chemical reaction?
-The stoichiometric ratio in a chemical reaction is the ratio of the amounts of reactants and products as written in the balanced chemical equation. It indicates how many moles of one substance react with a certain number of moles of another substance to form products.
How can you predict the mass of a product in a chemical reaction using stoichiometry?
-You can predict the mass of a product in a chemical reaction using stoichiometry by first converting the mass of the known reactant to moles, then using the balanced chemical equation to determine the stoichiometric ratio, and finally converting the expected number of moles of the product back to mass using the product's molar mass.
What is the molar mass of propane (C3H8)?
-The molar mass of propane (C3H8) is 44 grams per mole. This is calculated by adding the molar masses of three carbon atoms (3 x 12 amu) and eight hydrogen atoms (8 x 1 amu).
How does the concept of moles relate to the visibility of molecules in a chemical experiment?
-The concept of moles relates to the visibility of molecules in a chemical experiment by providing a way to work with a large number of molecules at once. Since individual molecules are too small to see, using moles allows chemists to handle and measure substances in quantities that are practical and observable.
Why is it important to ensure that units cancel out correctly during stoichiometric calculations?
-Ensuring that units cancel out correctly during stoichiometric calculations is important because it verifies that the calculation is dimensionally consistent and thus likely correct. It helps to avoid errors and ensures that the final answer is expressed in the desired units, such as grams or moles.
What is the final product mass of water expected from the combustion of 20 grams of propane, according to the script?
-The final product mass of water expected from the combustion of 20 grams of propane is 32 grams, as calculated using stoichiometric ratios and molar masses.
Outlines
🔍 Introduction to Moles in Chemistry
Professor Dave introduces the concept of moles, which is a fundamental unit in chemistry for counting molecules. He explains that while individual molecules are too small to see, they react in specific proportions by number, not mass. The mole, equivalent to Avogadro's number (6.022 x 10^23), provides a way to numerically represent a visible and manageable quantity of molecules. It bridges the gap between atomic mass units and grams, allowing chemists to perform stoichiometric calculations and predict outcomes of chemical reactions. The molar mass of a substance, expressed in grams, is also discussed, along with an example of calculating the mass of water produced from a combustion reaction involving propane.
📚 Stoichiometry and Mole Conversions
This paragraph delves into the process of performing stoichiometric calculations using moles. It emphasizes the importance of converting mass to moles and vice versa to predict chemical reaction outcomes. The example of calculating the expected mass of water from a given mass of propane is used to illustrate this process. The explanation includes how to use molar masses to convert between grams and moles, and how to apply stoichiometric ratios to find the expected product quantities. The paragraph concludes with a note on common problems involving unit conversions in chemistry and a prompt for viewers to subscribe for more tutorials and to reach out with any questions.
Mindmap
Keywords
💡Mole
💡Stoichiometry
💡Avogadro's number
💡Atomic mass units
💡Molar mass
💡Chemical reaction
💡Combustion reaction
💡Stoichiometric calculations
💡Balanced equations
💡Conversion factors
💡Periodic table
Highlights
A mole is a way to numerically represent a large number of molecules that we can observe and work with in chemistry.
Avogadro's number, 6.022 times ten to the 23rd, is the basis for the mole, representing the number of carbon atoms in 12 grams of carbon.
The mole serves as a bridge between atomic mass units and grams, allowing chemists to work with observable quantities.
Chemical reactions can be expressed in terms of moles, which simplifies the understanding of the proportions in which substances react.
Each substance has a molar mass, which is its molecular mass expressed in grams, allowing for the conversion between mass and moles.
For example, a mole of water molecules, which weigh 18 atomic mass units, corresponds to a mass of 18 grams.
Stoichiometric calculations using moles enable predictions about the outcomes of chemical reactions, such as the mass of products formed.
To perform stoichiometric calculations, one must convert the mass of a substance into moles using its molar mass.
The molar mass of propane is 44, and it can be used to convert grams of propane into moles for further calculations.
A four to one ratio in a chemical reaction, such as propane to water, can be determined by multiplying the moles of the reactant by the stoichiometric ratio.
The molar mass of water is used to find the mass of the expected product (water) from the number of moles calculated.
Units must be carefully managed during conversions to ensure they cancel out appropriately in stoichiometric calculations.
Common problems in chemistry involve converting mass to moles and back to mass, but for a different substance.
The process of converting between units using molar mass is demonstrated through an example calculation involving propane and water.
Mathematical predictions from stoichiometric calculations are fundamental to the application of chemistry in creating technology.
The importance of understanding moles and molar mass is emphasized for practical applications in chemistry.
The video provides a comprehensive tutorial on the concept of moles and their application in chemistry, encouraging further learning.
The audience is invited to subscribe for more tutorials and to reach out with questions, fostering an interactive learning environment.
Transcripts
Browse More Related Video

The mole and Avogadro's number | Atoms, compounds, and ions | Chemistry | Khan Academy

BTEC Applied Science: Unit 1 Chemistry The Mole
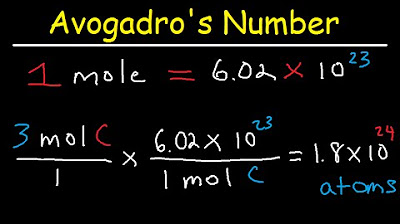
Avogadro's Number, The Mole, Grams, Atoms, Molar Mass Calculations - Introduction
Step by Step Stoichiometry Practice Problems | How to Pass Chemistry

GCSE Chemistry - The Mole (Higher Tier) #25

Concept of Mole | Avogadro's Number | Atoms and Molecules | Don't Memorise
5.0 / 5 (0 votes)
Thanks for rating: